Abstract
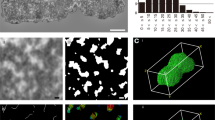
Changes in the morphology of human and murine chromosomes during the different stages of mitosis have been examined by scanning electron microscopy. Two important findings have emerged from this study. The first is that prophase chromosomes do not become split into pairs of chromatids until late prophase or early metaphase. This entails two distinct processes of condensation, the earlier one starting as condensations of chromosomes into chromomeres which then fuse to form a cylindrical body. After this cylindrical body has split in two longitudinally, further condensation occurs by mechanisms that probably include coiling of the chromatids as well as other processes. The second finding is that the centromeric heterochromatin does not split in two at the same time as the rest of the chromosome, but remains undivided until anaphase. It is proposed that the function of centromeric heterochromatin is to hold the chromatids together until anaphase, when they are separated by the concerted action of topoisomerase II acting on numerous similar sites provided by the repetitive nature of the satellite DNA in the heterochromatin. A lower limit to the size of blocks of centromeric heterochromatin is placed by the need for adequate mechanical strength to hold the chromatids together, and a higher limit by the necessity for rapid splitting of the heterochromatin at anaphase. Beyond these limits malsegregation will occur, leading to aneuploidy. Because the centromere remains undivided until anaphase, it cannot undergo the later stage of condensation found in the chromosome arms after separation into chromatids, and therefore the centromere remains as a constriction.
Similar content being viewed by others
References
Allshire RC, Cranston G, Gosden JR, Maule JC, Hastie ND, Fantes PA (1987) A fission yeast chromosome can replicate autonomously in mouse cells. Cell 50: 391–403
Bostock CJ, Summer AT (1978) The eukaryotic chromosome. North Holland, Amsterdam
Caserta M, Amadei A, diMauro E, Camilloni G (1989) In vitro preferential topoisomerization of bent DNA. Nucleic Acids Res 17: 8463–8474
Cooke CA, Heck MMS, Earnshaw WC (1987) The inner centromere protein (INCENP) antigens; movement from inner centromere to midbody during mitosis. J Cell Biol 105: 2053–2067
Darlington CD (1965) Cytology J & A Churchill, London
Eigsti OJ, Dustin P (1955) Colchicine—in agriculture, biology and chemistry. Iowa State College Press, Ames, Ch. 2
Flemming W (1880) Beitrag zur Kenntnis der Zelle und ihrer Lebenserscheinungen, Teil II. Archiv Mikrosk Anat 18: 151–259 (English translation in J Cell Biol 25: 1–69, 1965)
German J (1979) Roberts' syndrome. I. Cytological evidence for a disturbance in chromatid pairing. Clin Genet 16: 441–447
Golomb HM, Bahr GF (1974) Human chromatin from interphase to metaphase. Exp Cell Res 84: 79–87
Harrison CJ, Britch M, Allen TD, Harris R (1981) Scanning electron microscopy of the G-banded human karyotype. Exp Cell Res 134: 141–153
Harrison CJ, Jack EM, Allen TD (1987) Light and scanning electron microscopy of the same metaphase chromosomes. In: Hayat MA (ed) Correlative microscopy in biology: instrumentation and methods. Academic Press, New York, pp 189–248
Heslop-Harrison JS, Leitch AR, Schwarzacher T, Smith JB, Atkinson MD, Bennett MD (1989) The volumes and morphology of human chromosomes in mitotic reconstructions. Hum Genet 84: 27–34
Holm C, Stearns T, Botstein D (1989) DNA topoisomerase II must act at mitosis to prevent nondisjunction and chromosome breakage. Mol Cell Biol 9: 159–168
John B (1988) The biology of heterochromatin. In: Verma RS (ed) Heterochromatin. Cambridge University Press, Cambridge, pp 1–147
John B, Miklos GLG (1979) Functional aspects of satellite DNA and heterochromatin. Int Rev Cytol 58: 1–114
Lica LM, Narayanswami S, Hamkalo BA (1986) Mouse satellite DNA, centromere structure, and sister chromatid pairing. J Cell Biol 103: 1145–1151
Madan K, Lindhout D, Palan A (1987) Premature centromere division (PCD): a dominantly inherited cytogenetic anomaly. Hum Genet 77: 193–196
Manton I (1950) The spiral structure of chromosomes. Biol Rev 25: 486–508
Martinez-Balbas A, Rodriguez-Campos A, Garcia-Ramirez M, Sainz J, Carrera P, Aymami J, Azorin F (1990) Satellite DNAs contain sequences that induce curvature. Biochemistry 29: 2342–2348
Mazia D (1961) Mitosis and the physiology of cell division. In: Brachet J, Mirsky AE (eds) the cell, vol III. Academic Press, New York, pp 77–412
Mullinger AM, Johnson RT (1983) Units of chromosome replication and packing. J Cell Sci 64: 179–193
Mullinger AM, Johnson RT (1987) Disassembly of the mammalian metaphase chromosome into its subunits: studies with ultraviolet light and repair synthesis inhibitors. J Cell Sci 87: 55–69
Ohnuki Y (1968) Structure of chromosomes. I. Morphological studies of the spiral structure of human somatic chromosomes. Chromosoma 25: 402–428
Radic MZ, Lundgren K, Hamkalo B (1987) Curvature of mouse satellite DNA and condensation of heterochromatin. Cell 50: 1101–1108
Rieger R, Michaelis A, Green MM (1968) A glossary of genetics and cytogenetics, 3rd edn. George Allen & Unwin, London, Springer, Berlin
Röhme D, Heneen WK (1982) Banding patterns in prematurely condensed chromosomes and the underlying structure of the chromosome. In: Rao PN, Johnson RT, Sperling K (eds) Premature chromosome condensation. Academic Press, New York, pp 131–157
Rudd NL, Teshima IE, Martin SH, Sisken JE, Weksberg R (1983) A dominantly inherited cytogenetic anomaly: a possible cell division mutant. Hum Genet 65: 117–122
Shen CC, Shen C-KJ (1990) Specificity and flexibility of the recognition of DNA helical structure by eukaryotic topoisomerase I. J Mol Biol 212: 67–78
Sorsa V (1973) Condensation of chromosomes during mitotic prophase. Hereditas 75: 101–108
Spitzner JR, Muller MT (1988) A consensus sequence for cleavage by vertebrate DNA topoisomerase II. Nucleic Acids Res 16: 5533–5556
Sumner AT, Ross A (1989) Factors affecting preparation of chromosomes for scanning electron microscopy using osmium impregnation. Scanning Microsc [Suppl] 3: 87–99
Swanson CP (1963) Cytology and cytogenetics. Macmillan, London
Tomkins D, Hunter A, Roberts M (1979) Cytogenetic findings in Roberts-SC phocomelia syndrome(s). Am J Med Genet 4: 17–26
Vig BK (1987) Sequence of centromere separation: a possible role for repetitive DNA. Mutagenesis. 2: 155–159
Walker PMB (1971) “Repetitive” DNA in higher organisms. Prog Biophys Mol Biol 23: 145–190
White MJD (1961) The chromosomes, 5th edn. Methuen, London
Yanagida M (1989) Gene products required for chromosome separation. J Cell Sci [Suppl] 12: 213–229
Yunis JJ, Bahr GF (1979) Chromatin fibre organisation of human interphase and prophase chromosomes. Exp Cell Res 122: 63–72
Yunis JJ, Yasmineh WG (1971) Heterochromatin, satellite DNA, and cell function. Science 174: 1200–1209
Author information
Authors and Affiliations
Additional information
by U. Scheer
Rights and permissions
About this article
Cite this article
Sumner, A.T. Scanning electron microscopy of mammalian chromosomes from prophase to telophase. Chromosoma 100, 410–418 (1991). https://doi.org/10.1007/BF00337519
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00337519