Summary
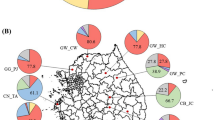
The diversity of Bradyrhizobium japonicum in agricultural fields has not been well characterized. Therefore a study was conducted to determine the serotypic diversity of B. japonicum both within and among six fields in the Coastal Plain and Piedmont of North Carolina where soybeans [Glycine max (L.) Merr.] are grown. Nodule samples were collected from non-inoculated standing soybean crops. Both nodules and isolates were typed by the enzyme-linked immunosorbent assay (ELISA) technique. Serotypes and their proportions varied both within and among locations. Common serotypes in order of abundance across all sites were 76, M1 (multiple reaction beyween 31 and 94), 94, 24, and 122, and together accounted for over 66% of the typable reactions. No cultivar effect on serotype distribution was observed. Unknown types ranged from 4 to 24%. Based on the total number of serotypes identified and the Shannon diversity index (H′), the mean population diversity was 0.76 for the Piedmont sites and 0.91 for the Coastal Plain sites.
Similar content being viewed by others
References
Abel GH, Erdman LH (1964) Response of Lee soybeans to different strains of Rhizobium japonicum. Agron J 56:423–424
Caldwell BE, Hartwig EE (1970) Serological distribution of root nodule bacteria in soils of the southeastern United States of America. Agron J 63:621–622
Caldwell BE, Vest G (1968) Nodulation interactions between soybean genotypes and serogroups of Rhizobium japonicum. Crop Sci 8:680–682
Caldwell BE, Vest G (1970) Effects of Rhizobium japonicum on soybean yields. Crop Sci 10:19–21
Caldwell BE, Weber DF (1970) Distribution of Rhizobium japonicum serogroups in nodules as affected by planting dates. Agron J 62:621–622
Crist DK, Wyza RE (1984) Preservation of Rhizobium viability and symbiotic infectivity by suspension in water. Appl Environ Microbiol 47:895–897
Darmighi SM, Frederick LR, Anderson JC (1967) Serogroups of Rhizobium japonicum in soybean nodules as affected by soil types. Agron J 59:10–12
Devine TE (1984) Genetics and breeding for nitrogen fixation. In: Alexander M (ed) Biological nitrogen fixation: Ecology, technology and physiology. Plenum Press, New York, pp 127–154
Elkins DM, Hamilton G, Chan CKY, Briskovich MA, Vandeventer JW (1976) Effects of cropping history on soybean growth and nodulation and soil rhizobia. Agron J 68:513–517
Fuhrmann J, Wollum AG II (1985) Simplified enzyme-linked immunosorbent assay for routine identification of Rhizobium japonicum antigens. Appl Environ Microbiol 49:1010–1013
Gibson AH, Dudman WF, Weaver RW, Horton JC, Anderson JC (1971) Variation within serogroup 123 of Rhizobium japonicum. Plant and Soil, Spec 35:33–37
Ham GE, Caldwell VB, Johnson HW (1971) Evaluation of Rhizobium japonicum inoculants in soils containing naturalized populations of rhizobia. Agron J 63:301–303
Holder-Franklin MA (1986) Ecological relationships of microbiota in water and soil as revealed by diversity measurements. In: Tate RL III (ed) Microbial autecology. John Wiley, New York, pp 93–132
Hollis AB, Kloos WE, Elkan GH (1981) DNA:DNA hybridization studies of Rhizobium japonicum and related Rhizobiaceae. J Gen Microbiol 123:215–222
Hunt PG, Matheny TA, Wollum AG II (1985) Rhizobium japonicum nodular occupancy, nitrogen accumulation and yield for determinate soybean under conservation and conventional tillage. Agron J 77:579–584
Johnson HW, Means UM (1960) Interactions between genotypes of soybeans and genotypes of nodulating bacteria. Agron J 52:651–654
Kempton RA (1979) The structure of species abundance and measurement of diversity. Biometrics 35:307–321
Kidby DP, Sandford P, Herman A, Cadmus M (1977) Maintenance procedures for the curtailment of genetic instability in Xanthomonas campestris NRRLB-1459. Appl Environ Microbiol 33:840–845
Kvien CS, Ham GE, Lambert JW (1981) Recovery of introduced Rhizobium japonicum strains by soybean genotypes. Agron J 73:900–905
Legendre L, Legendre P (1983) Numerical ecology. Elsevier, Amsterdam
Lindemann WC, Schmidt EL, Ham GE (1974) Evidence for double infection within soybean nodules. Soil Sci 118:274–279
Mahler RL, Wollum AG II (1982) Seasonal fluctuations of Rhizobium japonicum under a variety of field conditions in North Carolina. Soil Sci Soc Am J 134:317–324
May SN, Bohlool BB (1983) Competition among Rhizobium leguminosarum strains for nodulation of lentils (Lens esculenta). Appl Environ Microbiol 45:960–965
McClure PR, Israel DW (1979) Transport of nitrogen in xylem of soybean plants. Plant Physiol 64:411–416
Mehlich A (1953) Determination of P, Ca, Mg, K and Na. North Carolina Soil Test Division Memo, NC Dept Agricult, Raleigh
Mehlich A (1984) Photometric determination of humic matter in soils, a proposed method. Commun Soil Sci Plant Anal 15:1417–1422
Moawad HA, Schmidt EL (1987) Occurrence and nature of mixed infections in nodules of field-grown soybeans (Glycine max). Biol Fertil Soils 5:112–114
Munevar F (1981) Studies of the soybean-Rhizobium association as affected by high root temperature. Ph D thesis, North Carolina State University, Raleigh
Pielou EC (1975) Ecological diversity. John Wiley, New York
Routledge RD (1979) Diversity indices: Which ones are admissible? J Theor Biol 76:503–515
Semu E, Hume DJ, Corke CT (1979) Influence of soybean inoculation and nitrogen levels in populations and serogroups of Rhizobium japonicum in Ontario. Can J Microbiol 25:739–745
Speidel KL, Wollum AG II (1980) Leguminous inoculant quality: A manual. North Carolina Agric Res Serv Tech Bull no 260, Raleigh
Stanley J, Brown GG, Verma DPS (1985) Slow-growing Rhizobium japonicum comprises two highly divergent symbiotic types. J Bacteriol 163:148–154
State Soil Testing Laboratories (1984) Procedures Used by the State Soil Testing Laboratories in the Southern Region of the United States of America. Southern Cooperative Ser Bull 190. Agricultural Experiment Statem, Oklahoma State University, Stillwater
Vincent JM (1970) A manual for the practical study of root nodule bacteria. IBP Handbook no 15, Blackwell, Oxford
Weaver RW, Frederick LR, Dumenil LC (1972) Effect of cropping and soil properties on numbers of Rhizobium japonicum in Iowa soils. Soil Sci 114:137–141
Weber DF, Caldwell BE, Sloger C, Vest HG (1971) Some USDA studies on the soybean-Rhizobium symbiosis. Plant and Soil, Spec 35: 293–304
Weber DF, Miller VL (1972) Effect of soil temperature on Rhizobium japonicum serogroup distribution in soybean nodules. Agron J 64:796–798
Wilson JK (1930) Seasonal variation in the number of two species of Rhizobium in soil. Soil Sci 30:289–296
Author information
Authors and Affiliations
Additional information
Paper no. 12315 of the Journal Series of the North Carolina Agricultural Research Service, Raleigh, NC 27695-7643. The use of trade names in this publication does not imply endorsement by the North Carolina Agricultural Research Service of the products named or criticism of similar ones not mentioned
Rights and permissions
About this article
Cite this article
Mpepereki, S., Wollum, A.G. Diversity of indigenous Bradyrhizobium japonicum in North Carolina soils. Biol Fertil Soils 11, 121–127 (1991). https://doi.org/10.1007/BF00336376
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00336376