Abstract
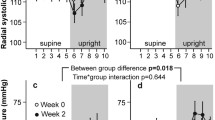
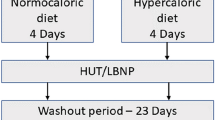
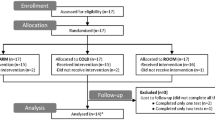
To evaluate influences on blood volume distribution, atrial natriuretic peptide concentrations (ANP) and thoracic and leg electrical impedance at 2.5 (TI2.5 and LI2.5, respectively) and 100 kHz (TI100 and LI100, respectively) were monitored during administration of ketanserin, noradrenaline and trimetaphan combined with lower body negative pressure (LBNP) in 12 subjects. Administration of clinically relevant doses of ketanserin alone did not induce changes in mean arterial pressure (MAP) or in the central blood volume, as electrical impedance and ANP concentrations did not change. During continued infusion of ketanserin an increase in MAP from a mean of 90 (range 83–108) to 113 (range 98–138) mmHg was induced by noradrenaline, but TI2.5 [mean 45.6 (range 39.3–54.2)] and TI100 [mean 33.8 (range 27.5–38.5) Ω] remainded stable until ganglionic blockade and LBNP were applied, when they increased by a mean of 3.1 (range 2.0–6.1) and 2.7 (range 1.1–4.2) Ω, respectively (P < 0.05). Conversely, LI2.5 [mean 79.6 (range 74.1–89.4)] and LI100 [mean 56.7 (range 52.4–63.3) Ω] decreased by a mean of 3.2 (range 1.2–8.0) and 2.3 (range 0.9–3.9) Ω ANP from a mean of 27.7 (range 10.2–62.7) to 12.7 (range 7.1–27.5) pmol· 1−1 and MAP fell to a mean of 62 (range 42–70) mmHg (P < 0.05). The heart rate was a mean of 75 (range 69–77) beats -min-' and did not change until LBNP, when it increased to a mean of 102 (range 78–104) beats · min−1, as presyncopal symptoms appeared. The data indicated that serotonergic blockade by ketanserin and α-sympathetic stimulation by noradrenaline did not affect blood volume distribution in normal humans, but that ganglionic blockade combined with LBNP reduced the central blood volume as leg volume increased; during central hypovolaemia tachycardia induced by ganglionic blockade did not prevent the fall in MAP, and thereby the appearance of presyncopal symptoms.
Similar content being viewed by others
References
Baker LE, Judy WV, Geddes LE, Langley FM, Hill DW (1971) The measurement of cardiac output by means of electrical impedance. Cardiovasc Res Cent Bull Houston 9: 135–145
Baylis PH, Stockley RA, Heath DA (1978) Influence of lower body negative pressure upon arginine vasopressin release. Clin Endocrinol 9: 89–95
Berman IR, Scheetz WL, Jenkins EB, Hufnagel HV (1971) Transthoracic electrical impedance as a guide to intravascular overload. Arch Surg 102: 61–64
Boer P, Roos JC, Geyskes GG, Dorhout Mees EJ (1979) Measurement of cardiac output by impedance cardiography under various conditions. Am J Physiol 237: H491-H496
Castenfors J, Sjöstrand T (1972) Circulatory control via vagal afferents. I. Adjustment of heart rate to variations of blood volume in the rat. Acta Physiol Scand 84: 347–354
Denniston JC, Maher JT, Reeves JT, Cruz JC, Cymerman A, Grover RF (1976) Measurement of cardiac output by electrical impedance at rest and during exercise. J Appl Physiol 40: 91–95
Ebert TJ, Smith JJ, Barney JA, Merrill DC, Smith GK (1986) The use of thoracic impedance for determining thoracic blood volume changes in man. Aviat Space Environ Med 57: 49–53
Edmunds AT, Godfrey S, Tolley M (1982) Cardiac output measured by transthoracic impedance cardiography at rest, during exercise and at various lung volumes. Clin Sci 63: 107–113
Ekman LG, Milson I, Arvidsson S, Biber B, Martinell S, Sjögvist BA (1990) Clinical evaluation of an ensemble-averaging impedance cardiograph for monitoring stroke volume during spontaneus breathing. Acta Anaesthesiol Scand 34: 190–196
Hill DW, Mohapatra SN, Welham KC, Stevenson ML (1976) The effect of a progressive decrease in the circulating blood volume of the dog on the transthoracic impedance. Eur J Intens Care Med 2: 119–124
Hinghofer-Szalkay H, Konig EM, Sauseng-Felleger G, Zambo-Polz C (1992) Biphasic blood volume changes with lower body suction in humans. Am J Physiol 263: 1270–1275
Jacobsen J, Hansen OB, Sztuk F, Warberg J, Knigge U, Secher NH (1993) Enhanced heart rate response to haemorrhage by ileus in the pig. Acta Physiol Scand 149: 293–301
Judy WV, Langley FM, McCowen KD, Stinnett DM, Baker LE, Johnson PC (1969) Comparative evaluation of the impedance and isotope dilution methods for measuring cardiac output. Aerosp Med 40: 532–536
Jónsson F, Madsen P, Jørgensen LG, Lunding M, Secher NH (1995) Thoracic electrical impedance and fluid balance during aortic surgery. Acta Anaesthesiol Scand 39: 513–517
Jørgensen LG, Perko M, Perko G, Secher NH (1993) Middle cerebral artery velocity during head-up tilt induced hypovolaemic shock in humans. Clin Physiol 13: 323–336
Larsen FF, Mogensen L, Tedner B (1987) Transthoracic electrical impedance at 1 and 100 kHz — a means for separating thoracic fluid compartments? Clin Physiol 7: 105–113
Loeppky JA, Kobayashi Y, Venters MD, Luft UC (1979) Effects of regional hemoconcentration during LBNP on plasma volume determinations. Aviat Space Environ Med 50: 763–767
Madsen P, Iversen H, Secher NH (1993) Central venous oxygen saturation during hypovolaemic shock in humans. Scand J Clin Lab Invest 53: 67–72
Matzen S, Knigge U, Schütten HJ, Warberg J, Secher NH (1990) Atrial natriuretic peptide during head-up tilt induced hypovolemic shock in man. Acta Physiol Scand 140: 161–166
Matzen S, Perko G, Groth S, Friedman DB, Secher NH (1991) Blood volume distribution during head-up tilt induced hypovolemic hypotension in man. Clin Physiol 11: 411–422
Matzen S, Secher NH, Pawelczyk JA, Perko G, Iversen H, Knigge U, Bach F, Warberg J (1993) Effect of serotonine receptor blockade on endocrine and cardiovascular responses to head-up tilt in humans. Acta Physiol Scand 149: 163–176
Muzi M, Ebert TJ, Tristani FE, Jeutter DC, Barney JA, Smith JJ (1985) Determination of cardiac output using ensemble-averaged impedance cardiograms. J Appl Physiol 58: 200–205
Murray RH, Thompson LJ, Bowers JA, Albright ChD (1968) Hemodynamic effects of graded hypovolemia and vasopressor syncope induced by lower body negative pressure. Am Heart J 68: 799–811
Patterson RP, Kubicek WG, Witsoe DA, From AHL (1978) Studies on the effect of controlled volume change on the thoracic electrical impedance. Med Biol Eng Comput 16: 531–536
Pawelczyk JA, Matzen S, Friedman DB, Secher NH (1994) Cardiovascular and hormonal responses to central hypovolaemia in humans. In: Secher NH, Pawelczyk JA, Ludbrook J (eds) Blood loss and hypovolaemic shock. Arnold, London, pp 25–36
Perko G, Perko MJ, Jansen E, Secher NH (1991) Thoracic impedance as an index of body fluid balance during cardiac surgery. Acta Anaesthesiol Scand 35: 568–571
Perko G, Payne G, Secher NH (1993) An indifference point for electrical impedance in humans. Acta Physiol Scand 148: 125–129
Perko G, Payne G, Linkis P, Jørgensen LG, Landow L, Warberg J, Secher NH (1994) Thoracic impedance and pulmonary atrial natriuretic peptide during head-up tilt induced hypovolaemic shock in humans. Acta Physiol Scand 150: 449–454
Perko G, Tilgreen R, Secher NH (1995) The venous pump does not affect the indifference point for electrical impedance in humans. Eur J Appl Physiol 72: 179–182
Reimann IW, Frolich JC (1983) Mechanisms of antihypertensive action of ketanserin in man. BMJ 287: 381–383
Sander-Jensen K, Mehlsen J, Stadeager C, Christensen NJ, Fahrenkrug J, Schwartz TW, Warberg J, Bie P (1988) Increase in vagal activity during hypotensive lower-body negative pressure in humans. Am J Physiol 255: R149-R156
Schütten HJ, Johannesen AC, Torp-Petersen C, Sander-Jensen K, Bie P, Warberg J (1987) Central venous pressure — a physiological stimulus for secretion of atrial natriuretic peptide in humans? Acta Physiol Scand 131: 265–272
Sheps DS, Petrovick ML, Kizakevich C, Wolfe C, Craige E (1982) Continuous noninvasive monitoring of left ventricular function during exercise by thoracic impedance cardiography: automated derivation of systolic time intervals. Am Heart J 103: 519–524
Shoemaker WC (1975) Pathophysiology and therapy of shock states. In: Walker WE, Taylor DE (eds) Intensive care. Churchill Livingstone, Edinburgh, pp 51–76
Smith JJ, Bush JE, Wiedmeier VT, Tristani FE (1970) Application of impedance cardiography to study of postural stress. J Appl Physiol 29: 133–11137
Thomasset A (1965) Electro-chemical volume measurement of extracellular fluid. Biophysic significance of impedance at one kilocycle. Lyon Med 214: 131–143
Van der Starre PJA, Reneman RS (1988) The alfa-adrenergic receptor blocking effect of ketanserin and the interaction between alfaadrenergic and S2-Serotonergic receptor blockade. J Cardiovasc Pharmacol 11 [Suppl 1]: S54-S61
Vanhoutte P, Amery A, Birkenhager W, Breckenridge A, Buehler F, Distler A (1988) Serotonergic mechanisms in hypertension. Focus on the effects of ketanserin. Hypertension 11: 111–133
Victor RG, Leimbach WN (1987) Effects of lower body negative pressure on sympathetic discharge to leg muscles in humans. J Appl Physiol 63: 2558–2562
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Perko, G., Schmidt, J.F., Warberg, J. et al. Pharmacological manipulation of cardiovascular responses to lower body negative pressure. Europ J Appl Physiol 73, 459–464 (1996). https://doi.org/10.1007/BF00334424
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00334424