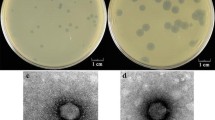
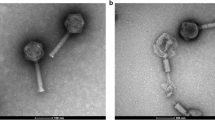
Summary
A gene for the lysin of Lactococcus lactis bacteriphage ΦvML3 was cloned using an Escherichia coli/bacteriophage lambda host-vector system. The gene was detected by its expression of antimicrobial activity against L. lactis cells in a bioassay. The cloned fragment was analysed by sub-cloning on to E. coli plasmid vectors and by restriction endonuclease and deletion mapping. Its entire DNA sequence was determined and an open reading frame for the lysin structural gene was identified. The sequenced lysin gene would express a protein of 187 amino acids with a molecular weight of 21090, which is in good agreement with that of a protein detected after in vitro transcription and translation of DNA encoding the gene. Expression of the lysin gene in E. coli and B. subtilis from an adjacent bacteriophage promoter was readily detected but in L. lactis expression of lysin was found to be lethal. The bacteriophage ΦvML3 lysin had sequence homology with protein 15 of B. subtilis bacteriophage PZA. This protein is involved in DNA packaging during bacteriophage muturation rather than in host cell lysin. The cloning and analysis of the ΦvML3 lysin gene is of importance in further understanding lactic streptococcal bacteriophages, for the development of positive selection vectors and for biotechnological applications of relevance to the dairy industry.
Similar content being viewed by others
References
Altman E, Young K, Garrett J, Altman R, Young R (1985) Subcelular localization of lethal lysis proteins of bacteriophages λ and Φ x 174. J Virol 53:1008–1011
Bachrach U, Friedmann A (1971) Practical procedures for the purification of bacterial viruses. Appl Microbiol 22:706–715
Bradford MM (1976) A rapid sensitive method for the quantification of microgram quantities of protein, utilizing the principle of protein dye binding. Anal biochem 12:248–254
Camacho A, Jiménez F, De la Torre J, Carrascosa JL, Mellado RP, Vásquez C, Vinuela E, Salas M (1977) Assembly of Bacillus subtilis phage Φ29. 1. Mutants in the cistrons coding for the structural proteins. Eur J Biochem 73:39–55
Carasscosa JL, Viñuela E, Salas M (1973) Proteins induced by Bacillus subtilis infected with bacteriophage Φ29. Virology 56:291–299
Casadaban MJ, Cohen SN (1980) Analysis of gene control signals by DNA fusion and cloning in Escherichia coli. J Mol Biol 138:179–207
Chang S, Cohen SN (1979) High frequency transformation of Bacillus subtilis protoplasts by plasmid DNA. Mol Gen Genet 168:111–115
Coveney JA, Fitzgerald GR, Daly C (1987) Detailed characterization and comparison of four lactic streptococcal bacteriophages based on morphology, restriction mapping, DNA homology and structural protein analysis. Appl Environ Microbiol 53:1439–1447
Davies FL, Gasson MJ (1984) Bacteriophages of dairy lactic-acid bacteria. In: Davies FL, Law BA (eds) Advances in the microbiology and biochemistry of cheese and fermented milk. Elsevier Applied Science Publishers, London, p 127
Davies FL, Underwood HM, Gasson MJ (1981) The value of plasmid profiles for strain identification in lactic streptococci and the relationship between Streptococcus lactis 712 ML3 and C2. J Appl Bacteriol 51:325–337
Delbrück M (1940) Adsorption of bacteriophage and lysis of the host. J Gen Physiol 23:643
De Vos WM (1987) Gene cloning and expression in lactic streptococci. FEMS Microbiol Rev 46:281–295
Fickett JW (1982) Recognition of protein coding regions in DNA sequences. Nucleic Acids Res 10:5303–5318
Gasson MJ (1983) Plasmid complements of Streptococcus lactis NCDO712 and other lactic streptococci after protoplast-induced curing. J Bacteriol 154:1–9
Geiduschek EP, Ito JP (1982) Regulatory mechanisms in the development of lytic bacteriophages in Bacillus subtilis. In: Dubnau DA (ed). The molecular biology of the bacilli, vol 1 Bacillus subtilis. Academic Press, New York, p 203
Hohn B (1979) In vitro packaging of λ and cosmid DNA. In: Wu R (Ed) Methods Enzymol 68:299–309
Hostomskŷ Z, Pačes V, Zedražil S (1985) Comparison of genomes of closely related phages Φ29, Φ15, and PZA using a rapid method of parallel physical mapping. FEBS Lett 188:123–126
Hoyt MA, Knight DM, Das A, Miller HI, Echols H (1982) Control of phage development by stability and synthesis of c11 protein: Role of the viral c111 and host hflA, himA and himD genes. Cell 31:565–573
Huyhn TV, Young RA, Davies RW (1985) Constructing and screening cDNA libraries in λgt10 and λgt11. In: Glover D (ed) DNA cloning techniques: A practical approach. IRL Press, Oxford, p 49
Jiménez F, Camacho A, De la Torre J, Viñuela E, Salas M (1977) Assembly of Bacillus subtilis phage Φ29. 2. Mutants in the cistrons coding for the non-structural proteins. Eur J Biochem 73:57–72
Karnik S, Billeter M (1983) Lysis function of RNA bacteriophage Qβ is mediated by the maturation (A2) protein. EMBO J 2:1521–1526
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Laemmli UK, Favre M (1973) Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol 80:575–599
Lennox ES (1955) Transduction of linked genetic characters of the host by bacteriophage P1. Virology 1:190–206
Loof M, Lembke J, Teuber M (1983) Characterization of the genome of the Streptococcus lactis “subsp. diacetylactis” bacteriophage P008 widespread in German cheese factories. Syst Appl Microbiol 4:13–423
Maeda S, Gasson MJ (1986) Cloning, expression and location of the Streptococcus lactis gene for phospho-β-cd-galactosidase. J Gen Microbiol 132:331–340
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Mata M, Ritzenthaler P (1988) Present state of lactic acid bacteria phage taxonomy. Biochimie 70:395–399
McLaughlin JR, Murray CL, Rabinowitz JC (1981) Unique features in ribosome binding site sequence of the gram-positive Staphylococcus aureus β-lactamase gene. J Biol Chem 255:8819–8830
Messing J (1983) New M13 vectors for cloning. Methods Enzymol 101:20–78
Moreno F, Camacho A, Viñuela E, Salas M (1974) Suppressor-sensitive mutants and genetic map of Bacillus subtilis Φ29. Virology 62:1–16
Norrander J, Kempe T, Messing J (1983) Construction of improved M13 vectors using oligodeoxynucleotide-directed mutagenesis. Gene 26:101–106
Oram JD, Reiter B (1965) Phage-associated lysins affecting group N and group D streptococci. J Gen Microbiol 40:57–70
Pačes V, Vlček Č, Urbanek P (1986) Nucleotide sequence of the late region of Bacillus subtilis phage PZA, a close relative of Φ29. Gene 44:107–114
Powell IB, Davidson BE (1985) Characterization of streptococcal bacteriophage c6A. J Gen Virol 66:2737–2741
Powell IB, Achen MG, Hillier AJ, Davidson BE (1988) A simple and rapid method for genetic transformation of lactic streptococci by electroporation. Appl Environ Microbiol 54:655–660
Reiter B, Oram JD (1963) Group N streptococcal phage lysin. J Gen Microbiol 32:29–32
Sanger F, Coulson AR, Barrell BG, Smith AJH, Roe BA (1980) Cloning in single-stranded bacteriophage as an aid to rapid DNA sequencing. J Mol Biol 143:161–178
Scalenghe F, Turco E, Edström JE, Pirrotta V, Melli M (1981) Microdissection and cloning of DNA from a specific region of Drosophila melanogaster polytene chromosomes. Chromosoma 82:205–216
Staden R (1980) A new computer method for the storage and manipulation of DNA gel reading data. NAR 8 (16):3673–3694
Terzaghi BK, Sandine NE (1975) Improve medium for lactic streptococci and their bacteriophages. Appl Microbiol 29:807–813
Teuber M, Lembke J (1983) The bacteriophages of lactic acid bacteria with emphasis on genetic aspects of group N lactic streptococci. Antonie van Leeuwenhoek 49:283–295
Twigg AJ, Sherrat DJ (1980) Trans-complementable mutants of ColE1. Nature 274:216–218
Van der Vossen JBM, van der Lelie D, Venema G (1987) Isolation and characterization of Streptococcus cremoris Wg2-specific promoters. Appl Environ Microbiol 53:2452–2457
Vieira J, Messing J (1982) The pUC plasmids, and M13mp7-d system for insertion mutagenesis and sequencing with synthetic universal primers. Gene 19:259–268
Weidel W (1951) Über die Zellmembran von E. coli β: Präparierung der Membranen gegenüber den Bakteriophagen. Z. Naturforschung 66:251
Weidel W (1958) Bacterial viruses (with particular reference to adsorption-penetration). Annu Rev Microbiol 12:27
Winter RB, Gold L (1983) Overproduction of bacteriophage Qβ maturation (A2) protein leads to cell lysis. Cell 33:877–885
Author information
Authors and Affiliations
Additional information
Communicated by H. Hennecke
Rights and permissions
About this article
Cite this article
Shearman, C., Underwood, H., Jury, K. et al. Cloning and DNA sequence analysis of a Lactococcus bacteriophage lysin gene. Molec Gen Genet 218, 214–221 (1989). https://doi.org/10.1007/BF00331271
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00331271