Summary
Cells defective in uracil-DNA glycosylase (ung:: Tn10) were used in two ways to reveal differences in select point mutations (GC to AT transitions) within the seven-tRNA operon of E. coli. The mutations were indicated as de novo or converted glutamine tRNA suppressor mutations in the genes glnU and/or glnV: (1) the kinetics of photoenzymatic monomerization of pyrimidine dimers quantitated by ung-dependent UV mutagenesis indicated more rapid repair of dimers at sites for converted suppressor mutation than of dimers at sites for de novo suppressor mutation, and (2) spontaneous deamination of cytosine was considerably more frequent at sites for converted suppressor mutation than at sites for de novo suppressor mutation. To explain these results we suggest the physical structure of the DNA in vivo is different at different sites in the seven-tRNA operon. The non-transcribed strand including specifically the anticodon region of the site for converted suppressor mutation may frequently be looped out in a single strand so that a T=C dimer is more accessible to DNA photolyase or a free cytosine residue of non-irradiated DNA is in an aqueous environment conducive to deamination. In addition, we analysed the spontaneous de novo suppressor mutation data to determine an estimate for the in vivo rate of cytosine deamination in double strand DNA of 3.2×1013/sec.
Similar content being viewed by others
References
Bachmann BJ (1983) Linkage map of Escherichia coli K-12, edition 7. Microbiol Rev 47:180–230
Bockrath RC, Palmer JE (1977) Differential repair of premutational UV-lesions at tRNA genes in E. coli. Mol Gen Genet 156:133–140
Bockrath R, Harper D, Kristoff S (1980) Crowding depression of UV-mutagenesis in Escherichia coli. Mutat Res 73:43–58
Bridges BA, Dennis RE, Munson RJ (1967a) Mutation in Escherichia coli B/r WP2 try by reversion or suppression of a chain terminating codon. Mutat Res 4:502–504
Bridges BA, Dennis RE, Munson RJ (1967b) Differential induction and repair of ultraviolet damages leading to true reversions and external suppressor mutations of an ochre codon in Escherichia coli B/r WP2. Genetics 57:89–908
Cheung M, Bockrath R (1970) On the specificity of UV mutagenesis in E. coli. Mutat Res 10:521–523
Duncan BK, Miller JH (1980) Mutagenic deamination of cytosine residues in DNA. Nature 287:560–561
Duncan BK, Weiss B (1982) Specific mutator effects of ung (uracil-DNA glycosylase) mutations in Escherichia coli. J Bacteriol 151:750–755
Engstrom J, Larsen S, Rogers S, Bockrath R (1984) UV-mutagenesis at a cloned target sequence: Converted suppressor mutation is insensitive to mutation frequency decline regardless of the gene orientation. Mutat Res 132:143–152
Fix D (1986) Thermal resistance of UV-mutagenesis to photoreactivation in E. coli B/r uvrA ung: estimate of activation energy and further analysis. Mol Gen Genet 204:452–456
Fix D, Bockrath R (1981) Thermal resistance to photoreactivation of specific mutations potentiated in E. coli B/r ung by ultraviolet light. Mol Gen Genet 182:7–11
Fix D, Bockrath R (1983) Targeted mutation at cytosine-containing pyrimidine dimers: Studies of E. coli B/r with acetophenone and 313 nm light. Proc Natl Acad Sci USA 80:4446–4449
Fix D, Glickman B (1986) Differential enhancement of spontaneous transition mutations in the lacI gene of an Ung- strain of Escherichia coli. Mutat Res (submitted)
Kato T, Shinoura Y, Templin A, Clark AJ (1980) Analysis of ultraviolet light-induced suppressor mutations in the strain Escherichia coli K-12 AB1157: An implication for molecular mechanism of UV mutagenesis. Mol Gen Genet 180:283–291
Kristoff S, Bockrath R (1983) Loss of photoreversibility for UV mutation in E. coli using 405 nm or near-UV challenge. Mutat Res 109:143–153
Lindahl T, Nyberg B (1974) Heat-induced deamination of cytosine residues in deoxyribonucleic acid. Biochemistry 13:3405–3410
Lindahl T (1979) DNA-glycosylases, endonucleases for apurinic/apyrimidinic sites, and base excision-repair. Prog Nucleic Acid Res Mol Biol 22:135–192
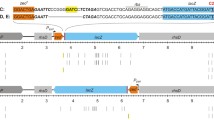
Nakajima N, Ozeki H, Shirmura Y (1981) Organization and structure of an E. coli tRNA operon containing seven tRNA genes. Cell 23:239–249
Osborn M, Person S (1967) Characterization of revertants of E. coli WU 36-10 and WP2 using amber mutants and an ochre mutant of bacteriophage T4. Mutat Res 4:404–407
Osborn M, Person S, Phillips SL, Funk F (1967) A determination of mutagen specificity in bacteria using nonsense mutants of bacteriophage T4. J Mol Biol 26:437–447
Ozeki H, Inokuchi H, Yamao F, Kodaira M, Sakano H, Ikemura T, Shimura Y (1980) Genetics of nonsense suppressor tRNAs in Escherichia coli. In: Soll D, Abelson JN, Schimmel PR (eds) Transfer RNA: biological aspects. Cold Spring Harbor Laboratory, pp 341–362
Person S, Osborn M (1968) The conversion of amber suppressors to ochre suppressors. Proc Natl Acad Sci USA 60:1030–1037
Sancar A, Rupp WD (1983) A novel repair enzyme: UvrABC excision nuclease of Escherichia coli cuts a DNA strand on both sides of the damaged region. Cell 33:249–260
Sancar GB, Smith FW, Sancar A (1985) Binding of Escherichia coli DNA photolyase to UV-irradiated DNA. Biochemistry 24:1849–1855
Rothwell MA, Green MHL, Bridges BA (1973) Genetic studies of class 2 nonsense suppressors in Escherichia coli. Genet Res (Camb) 22:233–237
Witkin EM (1966) Radiation-induced mutations and their repair. Science 152:1345–1353
Witkin EM (1976) Ultraviolet mutagenesis and inducible DNA repair in Escherichia coli. Bacteriol Rev 40:869–907
Wood RD (1985) Pyrimidine dimers are not the principal premutageneic lesions induced in lambda phage DNA by ultraviolet light. J Mol Biol 184:577–585
Author information
Authors and Affiliations
Additional information
Communicated by C. Auerbach
Rights and permissions
About this article
Cite this article
Bockrath, R., Mosbaugh, P. Mutation probe of gene structure in E. coli: suppressor mutations in the seven-tRNA operon. Molec Gen Genet 204, 457–462 (1986). https://doi.org/10.1007/BF00331024
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00331024