Summary
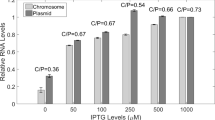
The lac operon shows anomalous expression in Proteus mirabilis: the maximal induced level is 10% or less of that in E. coli, while repression reduces this by a factor of only 2–5. We have sought to determine whether this effect relates in any way to CRP-mediated activation of expression, by comparing expression in P. mirabilis of lac operons (introduced for technical reasons on IncP1 plasmids) either regulatorily wild-type or bearing L8 or L8UV5. Derivatives of RP1 bearing L8UV5 were obtained by homogenotisation of pGC9114 (RP1:: Tn951) in a L8UV5 background; while derivatives of RP4 bearing lac +, L8 or L8UV5 were obtained by Mu-mediated translocation of chromosomal regions bearing these alleles, following partial heat-induction of Mucts62 on pGM14 (RP4:: Mucts62) in the appropriate hosts. These plasmids could be readily transferred to, and stably maintained in, the P. mirabilis strains employed. It was found that L8 reduced the maximal level of β-galactosidase activity, and L8UV5 restored this activity to around wild-type, in P. mirabilis quantitatively very much as in E. coli. Nevertheless, the low maximal level of expression and high basal level characteristic of the former host were unchanged. The simplest explanation of these results is that P. mirabilis contains a protein that mimics the E. coli CRP protein in interacting with the appropriate DNA binding site and thereby stimulating transcription; and that the anomalous regulation of lac in this host is unconnected with the CRP system.
Similar content being viewed by others
Abbreviations
- CPR=:
-
cyclic AMP receptor protein
References
Baumberg S, Cornelis G, Panagiotakopoulos M, Roberts M (1980) Expression of the lactose transposon Tn951 in Escherichia coli, Proteus and Pseudomonas. J Gen Microbiol 119:257–262
Baumberg S, Dennison S (1975) Variation in expression of sex factor genes in the Proteus-Providencia group relative to Escherichia coli. J Bacteriol 123:278–286
Baumberg S, Roberts M (1984) Anomalous expression of the E. coli lac operon in Proteus mirabilis. II. Effects of lacI and lacP mutations. Mol Gen Genet 198:166–171
Boucher B, Bergeron B, Barde de Bertalmio M, Dénarié J (1977) Introduction of bacteriophage Mu into Pseudomonas solanacearum and Rhizobium meliloti using the R factor RP4. J Gen Microbiol 98:253–263
Burkhardt HJ, Riess G, Pühler A (1979) Relationship of group P1 plasmids revealed by heteroduplex experiments: RP1, RP4, R68 and RK2 are identical. J Gen Microbiol 114:341–348
Colby C Jr, Hu ASL (1968a) The regulation of the synthesis of β-galactosidase in Proteus mirabilis F-lac. Biochim Biophys Acta 157:149–158
Colby C Jr, Hu ASL (1968b) Purification and comparison of β-galactosidase synthesized by Escherichia coli F-lac + and Proteus mirabilis F-lac +. Biochim Biophys Acta 157:167–177
Colby C Jr, Martin FD, Hu ASL (1968) Catabolite repression of the synthesis of β-galactosidase in Proteus mirabilis F-lac +. Biochim Biophys Acta 157:159–166
Cone KC, Sellitti MA, Steege DA (1983) lac repressor mRNA transcription terminates in vivo in the lac control region. J Biol Chem 258:11296–11304
Cornelis G, Ghosal D, Saedler H (1978) Tn951: a new transposon carrying a lactose operon. Mol Gen Genet 160:215–224
Cozzarelli NR (1980) DNA gyrase and the supercoiling of DNA. Science 207:953–960
Dale JW, Smith JT (1971) The purification and properties of the β-lactamase specified by the resistance factor R-1818 in Escherichia coli and Proteus mirabilis. Biochem J 123:493–500
Dénarié J, Rosenberg C, Bergeron B, Boucher C, Michel M, Barde de Bertalmio M (1977) Potential of RP4:: Mu plasmid for in vivo genetic engineering of gram negative bacteria. In: Bukhari AI, Shapiro J, Adhya S (eds) DNA insertion elements, plasmids and episomes. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, pp 507–520
Dennison S (1973) Control of fertility of resistance transfer factors in Proteus mirabilis and related bacterial species. Ph.D. thesis, University of Leeds
Dessein A, Schwartz M, Ullmann A (1978a) Catabolite repression in Escherichia coli mutants lacking cyclic AMP. Mol Gen Genet 162:83–87
Dessein A, Tillier F, Ullmann A (1978b) Catabolite modulator factor: physiological properties and in vivo effects. Mol Gen Genet 162:89–94
Faelen M, Toussaint A (1976) Bacteriophage Mu-1, a tool to transpose and to localize bacterial genes. J Mol Biol 104:525–539
Falkow S, Wohlhieter JA, Citarella RV, Baron LS (1964) Transfer of episomic elements to Proteus. II. Nature of lac + Proteus strains isolated from clinical speciments. J Bacteriol 88:1598–1601
Fried MG, Wu H-M, Crothers DM (1983) CAP binding to B and Z forms of DNA. Nucl Acids Res 11:2479–2494
Gemski P Jr, Wohlhieter JA, Baron LS (1967) Chromosome transfer between Escherichia coli and Proteus mirabilis. Proc Natl Acad Sci USA 58:1461–1467
Van Gijsegem F, Toussaint A, (1982) Chromosome transfer and R-prime formation by an RP4:: mini-Mu derivative in Escherichia coli, Salmonella typhimurium, Klebsiella pneumoniae, and Proteus mirabilis. Plasmid 7:30–44
Glesyna ML, Bolshakova TN, Gershanovitch VN (1983) Effect of ptsI and ptsH mutations on initiation of transcription of the Escherichia coli lactose operon. Mol Gen Genet 190:417–420
Greenblatt J, Li J, Adhya S, Friedman DI, Baron LS, Redfield B, Kung H-F, Weissbach H (1980) L factor that is required for β-galactosidase synthesis is the nusA gene product involved in transcription termination. Proc Natl Acad Sci USA 77:1991–1994
Guidi-Rontani C, Danchin A, Ullmann A (1980) Catabolite repression in Escherichia coli mutants lacking cyclic AMP receptor protein. Proc Natl Acad Sci USA 77:1991–1994
Kolb A, Buc H (1982) Is DNA unwound by the cyclic AMP receptor protein? Nucl Acids Res 10:473–485
Low B (1972) Escherichia coli K-12 F-prime factors, old and new. Bacteriol Rev 36:587–607
McKay DB, Steitz TA (1981) Structure of catabolite gene activator protein at 2.9 Å resolution suggests binding to left-handed B-DNA. Nature 290:744–749
Manson MD, Yanofsky C (1976) Tryptophan operon regulation in interspecific hybrids of enteric bacteria. J Bacteriol 126:679–689
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Morgan EA, Kaplan S (1976) Transcription of Escherichia coli ribosomal DNA in Proteus mirabilis. Mol Gen Genet 147:179–188
Morgan EA, Kaplan S (1977) Expression and stability of Escherichia coli F-prime factors in Proteus mirabilis. Mol Gen Genet 151:41–47
Pribnow D (1979) Genetic control signals in DNA. In: Goldberger RF (ed) Biological regulation and development, vol 1. Gene expression. Plenum, New York, pp 219–278
Primakoff P, Artz SW (1979) Positive control of lac operon expression in vitro by guanosine 5′-diphosphate 3′-diphosphate. Proc Natl Acad Sci USA 76:1726–1730
Reznikoff WS, Abelson JN (1980) The lac promoter. In: Miller JH, Reznikoff WS (eds) The operon (2nd ed). Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, pp 221–243
Roberts M (1982) Studies on the mechanism of anomalous expression of foreign genes in Proteus mirabilis. Ph.D.thesis, University of Leeds
Sanzey B (1979) Modulation of gene expression by drugs affecting deoxyribonucleic acid gyrase. J Bacteriol 138:40–47
Sanzey B, Ullmann A (1980) The effect of urea on catabolite sensitive operons in Escherichia coli K 12. Mol Gen Genet 178:611–616
Silverstone AE, Arditti RR, Magasanik B (1970) Catabolite-insensitive revertants of lac promoter mutations. Proc Natl Acad Sci USA 66:773–776
Smith JT (1969) R-factor expression in Gram-negative bacteria. J Gen Microbiol 55:109–120
Stubbs J, Horwitz A, Moses V (1973) Studies on β-galactoside transport in a Proteus mirabilis merodiploid carrying an Escherichia coli lactose operon. J Bacteriol 116:131–140
Ullmann A, Danchin A (1983) Role of cyclic AMP in bacteria. Adv Cylic Nucleotide Res 15:1–53
Ullmann A, Tillier F, Monod J (1976) Catabolite modulator factor: a possible mediator of catabolite repression in bacteria. Proc Natl Acad Sci USA 73:3476–3479
Ullmann A, Joseph E, Danchin A (1979) Cyclic AMP as a modulator of polarity in polycistronic transcriptional units. Proc Natl Acad Sci USA 76:3194–3197
Wohlhieter J, Falkow S, Citarella RV, Baron LS (1964) Characterization of DNA from a Proteus strain harboring an episome. J Mol Biol 9:576–588
Wohlhieter JA, Gemski P Jr, Baron LS (1975) Extensive segments of the Escherichia coli K12 chromosome in Proteus mirabilis diploids. Mol Gen Genet 139:93–101
Author information
Authors and Affiliations
Additional information
Communicated by C. Auerbach
Rights and permissions
About this article
Cite this article
Roberts, M., Baumberg, S. Anomalous expression of the E. coli lac operon in Proteus mirabilis . Molec Gen Genet 198, 159–165 (1984). https://doi.org/10.1007/BF00328716
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00328716