Summary
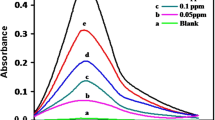
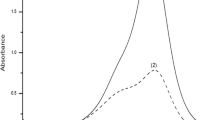
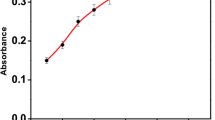
A new cationic dye, 1-(10-bromodecyl)-4-(4-aminonaphthylazo)-pyridinium bromide, was synthesized and evaluated as a new reagent for the determination of anionic surfactants. The reagent reacts with anionic surfactants, such as sodium dodecylsulphate and sodium dodecylbenzenesulphonate, to produce an ion associate in an aqueous medium. The colour change occurs simultaneously, and the colour development is very stable. This makes it possible to determine anionic surfactants directly by spectrophotometry without solvent extraction. The stoichiometric ratio of the ion associate was found to be 1:1 by the mole ratio method. The calibration graph was linear up to 2.5×10−6 mol/l. The apparent molar absorptivity of the ion associate was 5.3×104 l mol−1 cm−1 (at 595 nm). The relative standard deviation (n=10) for 1.2×10−6 mol/l sodium dodecylsulphate was 4.9%. The proposed method was applied to the determination of anionic surfactants in tap and river waters.
Similar content being viewed by others
References
Rand MC (1975) Standard methods for the examination of water and waste water. American Public Health Association, Washington, DC, pp 600–608
Japanese Industrial Standard, JIS K 0102, Japenese Standards Association, Tokyo, 1974
Tôei K, Miyata H, Motomizu S, Tetsumoto J (1978) Bunseki Kagaku 27:138–142
Motomizu S, Fujiwara S, Fujiwara A, Tôei K (1982) Anal Chem 54:392–397
Higuchi K, Shimoishi Y, Miyata H, Tôei K (1980) Analyst 105:768–773
Higuchi K, Shimoishi Y, Miyata H, Tôei K (1981) Chem Lett: 711–712
Higuchi K, Shimoishi Y, Miyata H, Tôei K (1982) Bull Chem Soc Jpn 55:621–622
Motomizu S, Oshima M, Kuroda T (1988) Analyst 113:747–753
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shimoishi, Y., Miyata, H. Spectrophotometric determination of anionic surfactants in tap and river waters with 1-(10-bromodecyl)-4-(4-aminonaphthylazo)-pyrimidinium bromide. Fresenius J Anal Chem 338, 46–49 (1990). https://doi.org/10.1007/BF00322783
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00322783