Summary
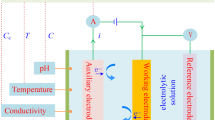
A simple galvanic cell without any external voltage for the trace determination of hydrogen sulphide is described. The signal corresponds to the concentration of the dissolved molecular H2S. For the calculation of total sulphide, the pH has to be taken into consideration. The sensor was tested in the range of concentration from 3·10−6 mol/l H2S to 9·10−5 mol/l H2S for pH<5. For pH 7.8 it covers a range from 1·10−6 mol/l H2S to 1·10−5 mol/l H2S. The sensitivity of the sensor substantially depends on the pH of the aqueous medium.
Similar content being viewed by others
References
Grasshoff K, Ehrhardt M, Kremling K (1983) Methods of Seawater Analysis. VCH, Weinheim
Rohde K-H, Nehring D (1979) Geod Geoph Veröff 4:24–27
Francom D, Goodwin L-R, Dieken F-P (1989) Anal Lett 22:2587–2600
Johnson K-S, Beehler C-L, Sakamoto-Arnold C-M (1986) Anal Chim Acta 179:245–257
Dobcnik D, Gomiscek D, Stergulec J (1990) Fresenius J Anal Chem 337:369–371
Frevert T, Galster H (1978) Schweiz Z Hydrol 40:199–208
Frevert T (1980) Schweiz Z Hydrol 42:255–268
Eckert W, Frevert T, Trüper H-G (1990) Water Res 24:1341–1346
Harsanyi E-G, Toth K, Pungor E (1984) Anal Chim Acta 161:333–341
Baumann E-W (1974) Anal Chem 46:1345–1347
Grünert A, Ballschmiter K, Tölg G (1968) Talanta 15:451–457
Jeroschewski P, Haase K, Trommer A, Gründler P (1993) Electroanalysis (in press)
Jeroschewski P, Söllig M, Berge H (1988) Z Chem 28:75
Jeroschewski P, Haase K, Söllig M (1988) DD 275924
Sedlak J-M, Blurton K-F (1976) Talanta 23:445–448
Stetter J-R, Sedlak J-M, Blurton K-F (1977) J Chromatogr Sci 15:125–128
Liss P-S, Slater P-G (1974) Nature 247:181–184
Spedding J, Vujcich M (1982) J Geophys Res 87:8853–8856
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jeroschewski, P., Haase, K., Trommer, A. et al. Galvanic sensor for the determination of hydrogen sulphide/sulphide in aqueous media. Fresenius J Anal Chem 346, 930–933 (1993). https://doi.org/10.1007/BF00322753
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00322753