Summary
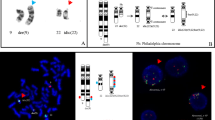
In a case of CML with a variant Philadelphia translocation (Ph1 or Ph) t (22;22) (q11;q13) in bone marrow cells and unstimulated peripheral blood cells, no cytogenetically detectable involvement of chromosome 9 was observed. Southern blot experiments using probes specific for bcr and c-sis however revealed rearrangement of the bcr, but not of PDGFB (c-sis) gene. Northern blot analysis of bone marrow RNA showed a very weak signal with the c-sis probe, while in a lymph-node biopsy PDGFB m-RNA could not be detected. Chromosomal in situ hybridization gave evidence for translocation of c-abl from chromosome 9 to Ph and of PDGFB from chromosome 22 to chromosome 9, as the result of a threefold translocation t(9;22;22).
Similar content being viewed by others
References
Abelson HT, Rabstein LS (1970) Lymphosarcoma: virusinduced thymic-independent disease in mice. Cancer Res 30: 2213–2222
Abramson S, Miller RG, Phillips RA (1977) The identification in adult bone marrow of pluripotent and restricted stem cells of the myeloid and lymphoid systems. J Exp Med 145: 1567–1579
Bartram CR, de Klein A, Hagemeijer A, van Agthoven T, Geurts van Kessel A, Bootsma D, Grosveld G, Ferguson-Smith MA, Davies T, Stone M, Heisterkamp N, Stephenson JR, Groffen J (1983) Translocation of c-abl oncogene correlates with the presence of a Philadelphia chromosome in chronic myelocytic leukemia. Nature 306: 277–280
Bartram CR, de Klein A, Hagemeijer A, Grosveld G, Heisterkamp N, Groffen J (1984) Localisation of the human c-sis oncogene in Ph1 positive and Ph1 negative chronic myelocytic leukemia by in situ hybridization. Blood 63: 223–225
Bartram CR, Anger B, Carbonell F, Kleihauer E (1985) Involvement of chromosome 9 in variant Ph1 translocation. Leuk Res 9: 1133–1137
Bernstein E (1988) Cytogenetics of chronic myelogenous leukemia. Semin Hematol 25: 20–34
Caspersson T, Gahrton G, Lindsten J, Zech L (1970) Identification of the Philadelphia chromosome on a number 22 by quinacrine mustard fluorescence analysis. Exp Cell Res 63: 238–240
Collins SJ, Kubonishi J, Miyoshi J, Groudine MT (1984) Altered transcription of the c-abl oncogene in K-562 and other chronic myelogenous leukemia cells. Science 225: 72–74
Dalla-Favera R, Gelman EP, Gallo RC, Wong-Staal F (1981) A human onc gene homologous to the transforming gene (v-sis) of simian sarcoma virus. Nature 292: 31–35
de Klein A, Geurts van Kessel A, Grosveld G, Bartram CR, Hagemeijer A, Bootsma D, Spurr NK, Heisterkamp N, Groffen J, Stephenson JR (1982) A cellular oncogene is translocated to the Philadelphia chromosome in chronic myelocytic leukemia. Nature 300: 765–767
Doolittle RF, Hunkapillar MW, Hood LE, Devare SG, Robbins KC, Aaronsson SA, Antoniades HN (1983) Simian Sarcoma Virus onc gene, v-sis, is derived from the gene (or genes) encoding a platelet-derived growth factor. Science 221: 275–277
Dreazen O, Canaani E, Gale RP (1988) Molecular biology of chronic myelogenous leukemia. Semin Hematol 25: 35–49
Falk M, Tesch H, Stein H, Diehl V, Jones DB, Fonatsch C, Bornkamm GW (1987) Phenotype versus immunoglobulin and T-cell receptor genotype of Hodgkin-derived cell lines: Activation of immature lymphoid cells in Hodgkin's disease. Int J Cancer 40: 262–26
Feinberg AP, Vogelstein B (1983) A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132: 6–13
Foerster W, Medau HJ, Löffler H (1974) Chronische myeloische Leukämie mit Philadelphia-Chromosom und Tandem-Translokation am 2. Chromosom Nr. 22; 46,XX, tan(22q+; 22q−). Klin Wochenschr 52: 123–126
Fonatsch C, Gradl G, Ragoussis J, Ziegler A (1987) Assignment of the TCP1 locus to the long arm of human chromosome 6 by in situ hybridization. Cytogenet Cell Genet 45: 109–112
Gale RP, Canaani E (1984) A 8-kilobase abl RNA transcript in chronic myelogenous leukemia. Proc Natl Acad Sci USA 81: 5648–5652
Goff SP, Gilboa E, Witte ON, Baltimore D (1980) Structure of the Abelson Murine Leukemia Virus genome and the homologous cellular gene: studies with cloned viral DNA. Cell 22: 777–785
Groffen J, Heisterkamp N, Stephenson JR, Geurts van Kessel A, de Klein A, Grosveld G, Bootsma D (1983) c-sis is translocated from chromosome 22 to chromosome 9 in chronic myelocytic leukemia. J Exp Med 158: 9–15
Groffen J, Stephenson JR, Heisterkamp N, de Klein A, Bartram CR, Grosveld G (1984) Philadelphia chromosomal breakpoints are clustered within a limited region, bcr, on chromosome 22. Cell 36: 93–99
Gyger M, Perreault C, Belanger R, Bonny Y (1985) Chronic myeloid leukemia with an unusual simple variant translocation: t(22;22) (q13;q11). Acta Haematol 73: 193–195
Hagemeijer A, Bartram CR, Smit EME, van Agthoven AJ, Bootsma D (1984) Is the chromosomal region 9q34 always involved in variants of the Ph1 translocation ?. Cancer Genet Cytogenet 13: 1–16
Hayata I, Kakati S, Sandberg AA (1973) A new translocation related to the Philadelphia chromosome. Lancet II: 1385
Heldin CH, Bäckström G, Östmann A, Hammacher A, Rönnstrand L, Rubin K, Nistér M, Westermark B (1988) Binding of different dimeric forms of PDGF to human fibroblasts: evidence for two separate receptor types. EMBO J 7: 1387–1393
Human Gene Mapping 9 (1987) Ninth International Workshop on Human Gene Mapping. Cytogenet Cell Genet, Volume 46
Julier C, Lathrop GM, Reghis A, Szajnert MF, Lalouel JM, Kaplan JC (1988) A linkage and physical map of chromosome 22, and some applications to gene mapping. Am J Hum Genet 42: 297–308
Maniatis T, Frisch EF, Sambrook J (1982) Molecular cloning — a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Mareni C, Sessarego M, Coviello DA, Origone P, Aimar F (1986) Involvement of chromosomal region 9q34 in a case of variant Ph1 translocation t(22;22). Leukemia Res 10: 1131–1137
Moos M, Gallwitz D (1983) Structure of two human β-actin related processed genes one of which is located next to a simple repetitive sequence. EMBO J 2: 757–761
Nowell PC, Hungerford DA (1960) A minute chromosome in human chronic granulocytic leukemia. Science 132: 1497
Prakash O, Yunis JJ (1984) High resolution chromosomes of the t(9;22) positive leukemias. Cancer Genet Cytogenet 11: 361–367
Reed KC, Mann DA (1985) Rapid transfer of DNA from agarose gels to nylon membranes. Nucleic Acids Res 13: 7207–7221
Robbins KC, Antoniades HN, Devare SG, Hunkapiller MW, Aaronson SA (1983) Structural and immunological similarities between simian sarcoma virus gene product(s) and human platelet-derived growth factor. Nature 305: 605–608
Rowley JD (1973) A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining. Nature 243: 290–291
Sandberg AA, Gemmill RM, Hecht BK, Hecht F (1986) Nomenclature: the Philadelphia chromosome or Ph without superscript. Cancer Genet Cytogenet 14: 1
Shtivelman E, Lifshitz B, Gale RP, Canaani E (1985) Fused transcript of abl and bcr genes in chronic myelogenous leukaemia. Nature 315: 550–554
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98: 503–517
Stam U, Heisterkamp N, Grosveld G, de Klein A, Verma RS, Coleman M, Dosik H, Groffen J (1985) Evidence of a new chimeric bcr/abl mRNA in patients with chronic myelocytic leukemia and the Philadelphia chromosome. N Engl J Med 313: 1429–1433
Stollmann B, Fonatsch C, Havers W (1985) Persistent Epstein-Barr virus infection associated with monosomy 7 or chromosome 3 abnormality in childhood myeloproliferative disorders. B J Haematol 60: 183–196
Strife A, Clarkson B (1988) Biology of chronic myelogenous leukemia: Is discordant maturation the primary defect? Semin Hematol 25: 1–19
Waterfield MD, Scrace T, Whittle N, Stroobant P, Johnsson A, Wasteson A, Westermark B, Heldin CH, Huang JS, Deuel TF (1983) Platelet-derived growth factor is structurally related to the putative transforming protein p28sis of simian sarcoma virus. Nature 304: 35–39
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gradl, G., Tesch, H., Schwieder, G. et al. Translocation of c-abl oncogene and PDGFB (c-sis) gene in a case of CML with 46, XY, t(22;22). Blut 58, 279–285 (1989). https://doi.org/10.1007/BF00320166
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00320166