Abstract
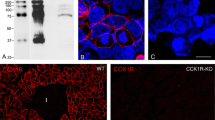
Electron-microscopic studies have revealed a heterogeneous distribution of gap junctions in the muscularis externa of mammalian intestines. This heterogeneity is observed at four different levels: among species; between small and large intestines; between longitudinal and circular muscle layers; and between subdivisions of the circular muscle layer. We correlated results obtained with two immunomethods, using an antibody to the known gap-junctional protein (connexin43) with ultrastructural findings, and further evaluated the respective sensitivity of these two approaches. For comparative reasons we also included the vascular smooth muscle of coronary arteries into our study. Two versions of the immunotechnique (peroxidase-antiperoxidase and fluorescence methods) were applied to frozen sections of murine, canine, and human small and large intestines, as well as to pig coronary artery. In the small intestine of all three species a very strong reactivity marked the outer main division of the circular muscle layer, while the longitudinal muscle layer as well as the inner thin division of the circular muscle layer were negative. In murine and human colon both muscle layers were negative, while in canine colon the border layer between the circular muscle and the submucosa reacted strongly, and scattered activity was found in the portion of the circular muscle layer (one tenth of its thickness) closest to the submucosa. The remainder of the circular muscle layer and the entire longitudinal muscle layer were negative in the canine colon. In the coronary artery we could not confirm the positive, specific labeling reported by other investigators (l.c.). In conclusion, we found close correlations at all four above-mentioned levels in the distribution of gap junctions in the gut musculature, as determined by binding of anticonnexin43 in comparison to conventional ultrastructural studies. Since no significant immunostaining was found in (i) the outer border of the circular muscle layer of the canine colon and (ii) the border layer between the submucosa and the circular muscle layer of human colon, where rare gap junctions have been identified at the ultrastructural level, we conclude that the electron-microscopic analysis is the more sensitive of the two methods.
Similar content being viewed by others
References
Bény JL, Connat JL (1992) An electron-microscopic study of smooth muscle cell dye coupling in the pig coronary arteries — Role of gap junctions. Circ Res 70:49–55
Berezin I, Huizinga JD, Daniel EE (1988) Interstitial cells of Cajal in the canine colon: a special communication network at the inner border of the circular muscle. J Comp Neurol 273:42–51
Berezin I, Huizinga JD, Daniel EE (1990) Structural characterization of interstitial cells of Cajal in myenteric plexus and muscle layers of canine colon. Can J Physiol Pharmacol 68:1419–1431
Beyer EC, Kistler J, Paul DL, Goodenough DA (1989) Antisera directed against connexin43 peptides react with a 43-kD protein localized to gap junctions in myocardium and other tissues. J Cell Biol 108:595–605
Chow E, Huizinga JD (1987) Myogenic electrical control activity in longitudinal muscle of human and dog colon. J Physiol (Lond) 392:21–34
Daniel EE, Duchon G, Henderson RM (1972) The ultrastructural bases for coordination of intestinal motility. Am J Dig Dis 17:289–298
Daniel EE, Daniel VP, Duchon G, Garfield RE, Nichols M, Malhotra SK, Oki M (1976) Is the nexus necessary for cell-to-cell coupling of smooth muscle. J Membr Biol 28:207–239
Dewey MM, Barr L (1962) Intercellular connection between smooth muscle cells: the nexus. Science 137:670–672
Faussone-Pellegrini MS, Cortesini C, Pantalone D (1990a) Neuromuscular structures specific to the submucosal border of the human colonic circular muscle layer. Can J Physiol Pharmacol 68:1437–1446
Faussone-Pellegrini MS, Pantalone D, Cortesini C (1990b) Smooth muscle cells, interstitial cells of Cajal and myenteric plexus interrelationships in the human colon. Acta Anat (Basel) 139:31–44
Gabella G, Blundell D (1979) Nexuses between the smooth muscle cells of the guinea pig ileum. J Cell Biol 82:239–247
Gabella G, Blundell D (1981) Gap junctions of the muscles of the small and large intestine. Cell Tissue Res 219:469–488
Garfield RE, Hertzberg EL (1990) Cell-to-cell coupling in the myometrium: Emil Bozler's prediction. Progr Clin Biol Res 327:673–681
Garfield RE, Thilander G, Blennerhassett MG, Sakai N (1992) Are gap junctions necessary for cell-to-cell coupling of smooth muscle?: an update. Can J Physiol Pharmacol 70:481–490
Hara Y, Kubota M, Szurszewski JH (1986) Electrophysiology of smooth muscle of the small intestine of some mammals. J Physiol (Lond) 372:501–520
Henderson RM, Duchon G, Daniel EE (1971) Cell contacts in duodenal smooth muscle layers. Am J Physiol 221:564–574
Huizinga JD, Chow E (1988) Electrotonic current spread in colonic smooth muscle. Am J Physiol 254:G702-G710
Huizinga JD, Stern HS, Chow E, Diamant NE, El-Sharkawy TY (1985) Electrophysiologic control of motility in the human colon. Gastroenterology 88:500–511
Huizinga JD, Shin A, Chow E (1988) Electrical coupling and pacemaker activity in colonic smooth muscle. Am J Physiol 255:C653-C660
Huizinga JD, Farraway L, Den Hertog A (1991) Effect of voltage and cyclic AMP on frequency of slow wave type action potentials in colonic smooth muscle. J Physiol (Lond) 442:31–45
Huizinga JD, Liu LWC, Blennerhassett MG, Thuneberg L, Molleman A (1992) Intercellular communication in smooth muscle. Experimentia 48:932–941
Liu LWC, Mikkelsen HB, Thuneberg L, Berezin I, Huizinga JD (1992) Electrical communication in areas with and without gap junctions in canine colon (abstract). Gastroenterology 103:1391
Mann JE Jr, Sperelakis N (1979) Further development of a model for electrical transmission between myocardial cells not connected by low-resistance pathways. J Electrocardiol 12:23–33
Mikkelsen HB, Rumessen JJ (1992) Characterization of macrophage-like cells in the external layers of human small and large intestine. Cell Tissue Res 270:273–279
Mikkelsen HB, Rumessen IT, Thuneberg L (1990) Prostaglandin H synthase immunoreactivity localized by immunoperoxidase technique (PAP) in the small intestine and kidney of rabbit and guinea-pig. Histochemistry 93:363–367
Mikkelson HB, Rumessen JJ, Qvortrup K (1991) Prostaglandin H synthase immunoreactivity in human gut. An immunohistochemical study. Histochemistry 96:295–299
Rumessen JJ, Thuneberg L (1991) Interstitial cells of Cajal in human small intestine. Ultrastructural identification and organization between the main smooth muscle layers. Gastroenterology 100:1417–1431
Rumessen JJ, Thuneberg L, Mikkelsen HB (1982) Plexus muscularis profundus and associated interstitial cells. II. Ultrastructural studies of mouse small intestine. Anat Rec 203:129–146
Rumessen JJ, Mikkelsen HB, Thuneberg L (1992) Ultrastructure of interstitial cells of Cajal associated with deep muscular plexus of human small intestine. Gastroenterology 102:56–68
Rumessen JJ, Peters S, Thuneberg L (1993) Light- and electron microscopical studies of interstitial cells of Cajal (ICC) and muscle cells at the submucosal border of human colon. Lab Invest 68:481–495
Sakai N, Tabb T, Garfield RE (1992) Studies of connexin-43 and cell-to-cell coupling in cultured human uterine smooth muscle. Am J Obstet Gynecol 167:1267–1277
Smith TK, Reed JB, Sanders KM (1987) Origin and propagation of electrical slow waves in circular muscle of canine proximal colon. Am J Physiol 252:C215-C224
Taylor AB, Kreulen D, Prosser CL (1977) Electron microscopy of the connective tissues between longitudinal and circular muscle of small intestine of cat. Am J Anat 150:427–441
Thuneberg L (1982) Interstitial cells of Cajal: intestinal pacemaker cells? Adv Anat Embryol Cell Biol 71:1–130
Thuneberg L (1989) Interstitial cells of Cajal. In: Schultz GS, Wood JD, Rauner BB (eds) Handbook of Physiology—The gastrointestinal system. American Physiology Society, Bethesda, USA, pp 349–386
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mikkelsen, H.B., Huizinga, J.D., Thuneberg, L. et al. Immunohistochemical localization of a gap junction protein (connexin43) in the muscularis externa of murine, canine, and human intestine. Cell Tissue Res 274, 249–256 (1993). https://doi.org/10.1007/BF00318744
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00318744