Abstract
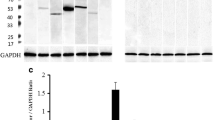
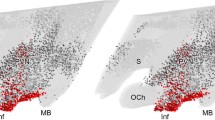
The regulation of cell activity, growth and metabolism by a number of growth factor receptors and proto-oncogene products involves tyrosine kinase activity resulting in autophosphorylation of the receptors and production of phosphorylated tyrosine-containing protein substrates. The identification and precise localization of phosphotyrosine (PY)-containing proteins are first steps in elucidating the functional role of tyrosine kinases in the modulation of the central nervous system and related areas. In the present report, we describe PY-containing proteins in the median eminence and adjacent pars tuberalis of the rat adenohypophysis by immunocytochemistry using light and electron microscopy, and by Western blotting analysis. PY-immunoreactivity was found to be most intense throughout the cytoplasm of a population of epithelial pars tuberalis cells. Polyacrylamide gel electrophoresis and Western blotting of tissue extracts from various brain and pituitary regions demonstrated a general pattern of 4 major bands of PY-proteins, with an additional dense band representing a 44 kDa protein that was highly phosphorylated on tyrosines and that was exclusively found in the pars tuberalis. Additional investigation for the presence of insulin receptors, a tyrosine kinase previously correlated with the distribution of PY-proteins, demonstrated a receptor localization in axons and nerve terminals in the external and internal zone of the median eminence. However, the large amount of different PY-proteins present in the secretory cell population of the pars tuberalis could not be attributed to the insulin receptor. Our findings demonstrate that there is a large amount of cell-specific tyrosine kinase activity in the median eminence and contacting the pars tuberalis; these may play a significant role for transduction of biological signals or metabolic regulation in the neuroendocrine region.
Similar content being viewed by others
References
Aguado F, Martinez-Murillo R, Sanchez-Franco F, Mellström B (1992) Distribution of insulin-like growth factor I receptor mRNA in rat brain. Relevance in the hypothalamo-hypophyseal system (abstract) Eur J Neurosci [Suppl 5]:306
Baker BL, Yu YY (1975) Immunocytochemical analysis of cells in the pars tuberalis of the rat hypophysis with antisera to hormones in the pars distalis. Cell Tissue Res 156:443–449
Baker BL, Karsch FJ, Hoffman DL, Beckman WC Jr (1977) The presence of gonadotropic and thyrotropic cells in the pituitary pars tuberalis of the monkey (Macaca mulatta). Biol Reprod 17:232–240
Baskin DG, Figlewicz DP, Woods SC, Porte D Jr, Dorsa DM (1987) Insulin in the brain. Annu Rev Physiol 49:335–347
Baskin DG, Wilcox BJ, Figlewicz DP, Dorsa DM (1988) Insulin and insulin-like growth factors in the CNS. Trends Neurosci 11:107–111
Baumert M, Takei K, Hartinger J, Burger PM, Mollard GF von, Maycox PR, De Camilli P, Jahn R (1990) p29: A novel tyrosine-phosphorylated membrane protein present in small clear vesicles of neurons and endocrine cells. J Cell Biol 110:1285–1294
Bergmann M, Wittkowski W, Hoffmann K (1989) Ultrastructural localization of thyrotropin (TSH)-like immunoreactivity in specific secretory cells of the hypophyseal pars tuberalis in the Djungarian hamster, Phodopus sungorus. Cell Tissue Res 256:649–652
Bohannon NJ, Figlewicz DP, Corp ES, Wilcox BJ, Porte D Jr, Baskin DG (1986) Identification of binding sites for an insulin-like growth factor (IGF-1) in the median eminence of the rat brain by quantitative autoradiography. Endocrinology 119:943–945
Bohannon NJ, Corp ES, Wilcox BJ, Figlewicz DP, Dorsa DM, Baskin DG (1988) Localization of binding sites for insulin-like growth factor-I (IGF-I) in the rat brain by quantitative autoradiography. Brain Res 444:205–213
Corp ES, Woods SC, Porte D Jr, Dorsa DM, Figlewicz DP, Baskin DG (1986) Localization of 125I-Insulin binding sites in the rat hypothalamus by quantitative autoradiography. Neurosci Lett 70:17–22
Dellmann HD, Stoeckel ME, Hindelang-Gertner C, Porte A, Stutinsky F (1974) A comparative ultrastructural study of the pars tuberalis of various mammals, the chicken and the newt. Cell Tissue Res 148:313–329
Ellis PD, Bissoon N, Gurd JW (1988) Synaptic protein tyrosine kinase: partial characterization and identification of endogenous substrates. J Neurochem 51:611–620
Girod C, Dubois MP, Trouillas J (1980) Immunohistochemical study of the pars tuberalis of the adenohypophysis in the monkey, Macaca irus. Cell Tissue Res 210:191–203
Hanley MR (1988) Proto-oncogenes in the nervous system. Neuron 1:175–182
Kahn CR, White MF (1988) The insulin receptor and the molecular mechanism of insulin action. J Clin Invest 82:1151–1156
Kamps MP (1991) Generation and use of anti-phosphotyrosine antibodies for immunoblotting. Methods Enzymol 201:101–110
Kobayashi T, Kobayashi T, Yamamoto K, Inatomi M (1963) Electron microscopy observation on the hypothalamo-hypophyseal system in the rat. I. The ultrafine structure of the contact region between the external layer of the infundibulum and pars tuberalis of the anterior pituitary. Endocrinol Jpn 10:69–80
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T. Nature 227:680–685
Lesniak MA, Hill MA, Kiess W, Rojeski M, Pert CB, Roth J (1988) Receptors for insulin-like growth factors I and II: autoradiographic localization in rat brain and comparison to receptors for insulin. Endocrinology 123:2089–2099
Luschka H (1860) Der Hirnanhang und die Steißdrüse des Menschen. Springer, Berlin
Marks JL, Porte D Jr, Baskin DG (1991) Localization of type I insulin-like growth factor receptor messenger RNA in the adult rat brain by in situ hybridization. Mol Endocrinol 5:1158–1168
Merchenthaler I (1991) Current status of brain hypophysiotropic factors-morphological aspects. TEM 2:219–226
Mitchison T, Kirschner M (1988) Cytoskeletal dynamics and nerve growth. Neuron 1:761–772
Morgan PJ, Williams LM, Davidson G, Lawson W, Howell E (1989) Melatonin receptors on ovine pars tuberalis: characterization and autoradiographical localization. J Neuroendocrinol 1:1–4
Moss AM, Unger JW, Moxley RT, Livingston JN (1990) Location of phosphotyrosine-containing proteins by immunocytochemistry in the rat forebrain corresponds to the distribution of the insulin receptor. Proc Natl Acad Sci USA 87:4453–4457
Nitsch C, Riesenberg R (1988) Immunocytochemical demonstration of GABAergic synaptic connections in rat substantia nigra after different lesions of the strionigral projection. Brain Res 461:127–142
Ordronneau P, Petrusz P (1980) Immunocytochemical demonstration of anterior pituitary hormones in the pars tuberalis of long-term hypophysectomized rats. Am J Anat 158:491–506
Pyper JM, Bolen JB (1990) Identification of a novel neuronal c-src exon expressed in human brain. Mol Cell Biol 10:2035–2040
Rosen OM (1989) Structure and function of insulin receptors. Diabetes 38:1508–1511
Schulze-Bonhage A, Wittkowski W (1990) Age-dependent changes of ultrastructure and thyroid-stimulating hormone immunore-activity in the pars tuberalis of the rat. Acta Anat 139:134–140
Sternberger LA (1979) Immunocytochemistry, 2nd edn. Wiley, New York
Sugrue MM, Bruge JS, Marshak DR, Greengard P, Gustafson EL (1990) Immunocytochemical localization of the neuron-specific form of the c-src gene product, pp60c-src(+), in rat brain. J Neurosci 10:2513–2527
Tilney F (1913) An analysis of the juxtaneural epithelial portion of the hypophysis cerebri with an embryological and histological account of an hitherto undescribed part of the organ. Anat Physiol 30:258–293
Ullrich A, Schlessinger J (1990) Signal transduction by receptors with tyrosine kinase activity. Cell 61:203–212
Unger J, McNeill TH, Moxley III RT, White M, Moss A, Livingston JN (1989) Distribution of insulin receptor-like immunoreactivity in the rat forebrain. Neuroscience 31:143–157
Unger JW, Moss AM, Livingston JN (1991a) Immunohistochemical localization of insulin receptors and phosphotyrosine in the brainstem of the adult rat. Neuroscience 42:853–861
Unger JW, Livingston JN, Moss AM (1991b) Insulin receptors in the central nervous system: localization, signalling mechanisms and functional aspects. Prog Neurobiol 36:343–362
Van Houten M, Posner BI (1981) Insulin receptors in the central nervous system: localization and characterization. In: Andreani D, De Pirro R, Lauro R, Olefsky J, Roth J (eds) Current views on insulin receptors, vol 41. Serono Symposia. Academic Press, London New York, pp 75–90
Van Houten M, Posner BI, Kopriwa BM, Brawer JR (1989) Insulin binding sites localized to nerve terminals in rat median eminence and arcuate nucleus. Science 207:1081–1083
Wagner KR, Mei L, Huganir RL (1991) Protein tyrosine kinases and phosphatases in the nervous system. Curr Opin Neurobiol 1:65–74
Warren SL, Nelson J (1987) Nonmitogenic morphoregulatory action of pp60v-src on multicellular epithelial structures. Mol Cell Biol 7:1326–1337
Watson RE Jr, Wiegand SJ, Clough RW, Hoffman GE (1986) Use of cryoprotectant to maintain long-term peptide immunoreactivity and tissue morphology. Peptides 7:155–159
White MR, Backer JM (1991) Preparation and use of anti-phosphotyrosine antibodies to study structure and function of insulin receptors. Methods Enzymol 201:65–79
Wiestler OD, Walter G (1988) Developmental expression of two forms of pp60c-src in mouse brain. Mol Cell Biol 8:502–504
Williams LM, Morgan PJ (1988) Demonstration of melatonin binding sites in the pars tuberalis of the rat. J Endocrinol 119:R1–3
Wittkowski WH, Schulze-Bonhage AH, Böckers TM (1992) The pars tuberalis of the hypophysis: a modulator of the pars distalis? Acta Endocrinol 126:285–290
Yarden Y, Ullrich A (1988) Growth factor receptor tyrosine kinases. Annu Rev Biochem 57:443–478
Author information
Authors and Affiliations
Additional information
This paper is dedicated to Professor Dr. Leonhardt on the occasion of his 75th birthday
Rights and permissions
About this article
Cite this article
Unger, J.W., Moss, A.M. & Livingston, J.N. The hypophyseal pars tuberalis is enriched with distinct phosphotyrosine-containing proteins not detected in other areas of the brain and pituitary. Cell Tissue Res 272, 499–507 (1993). https://doi.org/10.1007/BF00318556
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00318556