Abstract
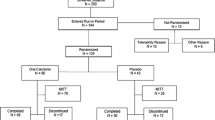
The objective of this study was to test the efficacy and safety of salmon calcitonin (sCT) suppository in postmenopausal women with previous hip fractures as an inhibitory agent of bone loss. The study was a single blind, randomized, and placebo-controlled trial comparing three parallel groups of patients. Fifty-four healthy women were randomly allocated to 1 year's treatment with either sCT 100 IU/6 times a week, 200 IU/3 times a week, or placebo/6 times a week. All groups received a calcium supplement of 500 mg daily. Fifteen patients left the study before its end, six of those due to adverse events, such as abdominal and rectal pain, nausea, headache, and diarrhea. Bone mineral density of the spine and the femoral neck was measured every 26 weeks, and biochemical markers of bone turnover were measured at baseline and week 12, 26, and 52. There were no significant changes in bone mineral density in the spine and in the hip in any of the treatment groups. No significant changes were observed in serum alkaline phosphatase, serum osteocalcin, urine hydroxyproline, and urine pyridinoline or deoxypyridinoline. Conclusively, we did not observe any significant effect on bone metabolism in women with postmenopausal osteoporosis after 1 year of treatment with sCT suppositories at the doses used.
Similar content being viewed by others
References
Marie PJ, Caulin F (1986) Mechanism underlying the effects of phosphate and calcitonin on bone histology in postmenopausal osteoporosis. Bone 7:17–22
Gruber HE, Ivey JL, Baylink DJ, Matthews M, Nelp WB, Sisom K, Chesnut CH (1984) Long-term calcitonin therapy in postmenopausal osteoporosis. Metabolism 33:295–303
Overgaard K, Hansen MA, Jensen SB, Christiansen C (1992) Effect of salcatonin given intranasally on bone mass and fracture rates in established osteoporosis: a dose-response study. Br Med J 305:556–561
Reginster JY, Denis D, Albert A, Deroisy R, Lecart MP, Fontaine MA, Lambelin P, Franchimont P (1987) 1-year controlled randomized trial of prevention of early postmenopausal bone loss by intranasal calcitonin. Lancet 2:1481–1483
Overgaard K, Riis BJ, Christiansen C, Hansen MA (1989) Effect of salcatonin given intranasally on early postmenopausal bone loss. Br Med J 299:477–479
Roth A, Kolaric K (1986) Analgetic activity of calcitonin in patients with painful osteolytic metastasis of breast cancer. Oncology 43:45–51
Thamsborg G, Storm TL, Brinch E, Sykulski R, Fogh-Andersen N, Nistrup HS, Sørensen OH (1990) The effect of different doses of nasal calcitonin on plasma cyclic AMP and serum ionized calcium. Calcif Tissue Int 46:5–8
Buclin T, Randin JP, Jacquet AF, Azria M, Attinger M, Gomez F, Burckhardt P (1987) The effect of rectal and nasal administration of salmon calcitonin in normal subjects. Calcif Tissue Int 47:252–258
Nagant de Deuxchaisnes C, Devogelear JP, Huaux JP, Dufour JP, Esselinckx W, Engelbeen JP, Stasse P, Hermans P, Buisseret JP (1987) New modes of administration of salmon calcitonin in Paget's disease. Clin Orthop Rel Res 217:56–71
Thiébaud D, Burckhardt P, Jaeger Ph, Azria M (1987) Effectiveness of salmon calcitonin administered as suppositories in tumor-induced hypercalcemia. Am J Med 82:745–750
Angelini GP, Gonelli S, Palmieri R, Annusdei D, Vassellatti D, Gennari C (1992) Effect of salmon calcitonin given by suppositories on bone mass in osteoporotic patients: a double blind placebo-controlled study. Bone Miner 16 (suppl 1):173
Overgaard K, Hansen MA, Dirksen KL, Christiansen C (1992) Rectal salmon calcitonin for the treatment of postmenopausal osteoporosis. Calcif Tissue Int 51:184–188
Nielsen KH, Brixen K, Kassem M, Charles P, Mosekilde L (1992) Inhibition of the morning cortisol peak abolishes the expected morning decrease in serum osteocalcin in normal males: evidence of a controlling effect of serum cortisol on the circadian rhythm in serum osteocalcin 6:1410–1414
Kollerup G, Thamsborg G, Bhatia H, Sørensen OH (1992) Quantitation of urinary hydroxypyridinium cross-links from collagen by high-performance liquid chromatography. Scan J Clin Lab Invest 52:657–662
Kivirikko KI, Laitinen O, Prockop DJ (1967) Modification of a specific assay for hydroxyproline in urine. Anal Biochem 19:249–255
Civitelli R, Gonnelli S, Zacchei F, Bigazzi S, Vattimo A, Avioli LV, Gennari C (1988) Bone turnover in post-menopausal osteoporosis. J Clin Invest 82:1268–1274
Delmas PD, Schlemmer A, Gineyts E, Riis BJ, Christiansen C (1991) Urinary excretion of pyridinoline cross-links correlates with bone turnover measured on iliac crest biopsy in patient with vertebral osteoporosis. J Bone Miner Res 6:639–644
Eastell R, Hamton L, Colwell A, Green JR (1990) Urinary cross-links are highly correlated with radioisotopic measurements of bone resorption. In: Christiansen C, Overgaard K (eds) Osteoporosis 1990. Osteopress Aps, Copenhagen, pp 469–470
Avioli LV (1991) Heterogeneity of osteoporotic syndromes and the response to calcitonin therapy. Calcif Tissue Int 49 (suppl 2S): 16–19
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kollerup, G., Hermann, A.P., Brixen, K. et al. Effects of salmon calcitonin suppositories on bone mass and turnover in established osteoporosis. Calcif Tissue Int 54, 12–15 (1994). https://doi.org/10.1007/BF00316282
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00316282