Summary
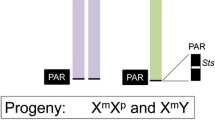
The Sxr (sex reversed) factor in the mouse described by Cattanach et al. (1971) causes sex reversal of genetic female mice, thus producing XX males. Its primary effect is induction of testicular development in XX embryos.
In the present study, the early steps of testicular development in sex reversed XX and in XY mouse embryos are examined. The embryos are bred from male carriers of the Sxr mutation. After determination of the sex chromosomes from chromosomal spreads, the appearance of testis cords is evaluated in sex reversed XX embryos and their XY littermates at 12 1/2 and 13 days of pregnancy. In sex reversed XX embryos testis cords appear at the same stage of development as in XY embryos.
From day 13 to 17 of pregnancy, the volume of the testis anlage is determined by planimetry in sex reversed XX and in normal XY embryos from separate litters. No significant difference is found in the growth rate of XX and XY testes. Measurements of the diameter of the testis cords also indicate the same developmental pattern in both groups. Thus, during induction of a testis the Sxr factor perfectly imitates the Y chromosome in all parameters investigated.
Similar content being viewed by others
References
Brambell, G.W.R.: The development and morphology of the gonads of the mouse. Part 1: The morphogenesis of the indifferent gonad and of the ovary. Proc. Soc. Lond. Ser. B, 101, 391–409 (1927)
Cattanach, B.M.: Sex reversal in the mouse and other mammals. In: The early development of mammals (M. Balls, A.E. Wild, eds.), pp. 305–317. Cambridge 1975
Cattanach, B.M., Pollard, C.E., Hawkes, S.G.: Sex reversed mice: XX and XO males. Cytogenetics 10, 318–337 (1971)
Chapelle De La, A.: Analytic review: Nature and origin of males with XX sex chromosomes. Amer. J. Hum. Genet. 24, 71–105 (1972)
Chung, K.W.: A morphological and histochemical study of Sertoli cells in normal and XX sex reversed mice. Am. J. Anat. 139, 369–388 (1974)
Chung, K.W., Blackburn, W., Bullock, L., Ohno, S., Bardin, C.W.: Testicular structure-function relationships in the mouse with XX sex reversal. Anat. Rec. 172, 290 (Abstract) (1972)
Drews, U., Drews, U.: Regression of mouse mammary gland anlagen in recombinants of Tfm and wildtype tissues: Testosterone acts via the mesenchyme. Cell, 10, 401–404 (1977)
Eaton, O.N.: The relation between Polled and hermaphroditic characters in dairy goats. Genetics 30, 51–61 (1945)
Evans, E.P., Burtenshaw, M.D., and Ford, C.E.: Chromosomes of mouse embryos and newborn young: preparations from membranes and tail tip. Stain Technol. 47, 229–234 (1972)
Hamerton, J.L., Dickson, J.M., Pollard, C.E., Grieves, S.A., Short, R.V.: Genetic intersexuality in goats. J. Reprod. Fert., Suppl. 7, 25–51 (1969)
Jean, C. Volumetric development of gonads in mouse embryos. C.R. Soc. Biol. 165 2341–2345 (1971)
Jost, A., Vigier, B., Prépin, J., Perchellet, J.P.: Studies on sex differentiation in mammals. Recent Progr. Hormone Res. 29, 1–39 (1973)
Kasdan, R., Nankin, H.R., Troen, P., Wald, M., Pan, S., Yanaihasa, T.: Paternal transmission of maleness in XX human beings. New Eng. J. Med. 288, 539–545 (1973)
Muschge, H.E.: Histometrische Untersuchungen am Rattenhoden nach Hypophysektomie und nach Choriongonadotropinzufuhr. Endokrinologie 30, 281–294 (1953)
Ohno, S.: Simplicity of mammalian regulatory systems inferred by single gene determination of sex phenotypes. Nature 234, 134–137 (1971)
Ohno, S., Nagai, Y., Caccarese, S.: Testicular cells lysostripped of H-Y antigen organize ovarian folliclelike aggregates Cytogenet. Cell Genet. 20, 351–364 (1978)
Wachtel, St.S., Ohno, S., Koo, G.C., Boyse, E.A.: Possible role for H-Y antigen in the primary determination of sex. Nature 257, 235–236 (1975)
Wagenen, G. Van, Simpson, M.E.: Embryology of the ovary and testis Homo sapiens and Macaca mulatta. New Haven and London: Yale University Press 1965
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Spoljar, M., Drews, U. Identical action of Y chromosome and Sxr factor in early testicular development in the mouse. Anat Embryol 155, 115–126 (1979). https://doi.org/10.1007/BF00315736
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00315736