Summary
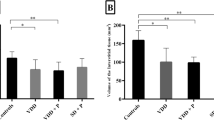
Differences in the thymus of young and old male CSE Wistar rats were examined by use of routine histological stains on paraffin-embedded sections. There was a highly significant loss of thymic weight and disruption of architecture with age. Both surgical castration and chemical castration induced by a luteinizing hormone-releasing hormone analogue (Goserelin) caused a significant increase in thymic weight and the reappearance of a well-defined cortex and medulla in ageing rats. Cell surface antigens were detected on cryosections after incubation with a range of monoclonal antibodies. The Pan T cell marker (detected with antibody W3/13) showed fewer positive cells in ageing rats, and an increase after chemical castration. The smaller glands of old rats had fewer positive T cells with CD4 (MRC OX35) and CD8 (MRC OX8) antigens, and more after chemical castration in both young and ageing rats, but the greatest changes were seen in the intensity of Class II major histocompatibility complex (MRC OX6) immunoreactivity. In both young and ageing chemically-castrated rats, the numbers of cells and the intensity of immunoreactivity were greatly increased in the medulla.
Similar content being viewed by others
References
Andersen DH (1932) The relationship between the thymus and reproduction. Physiol Rev 12:1–22
Bach MA, Beaurain G (1979) Respective influence of extrinsic and intrinsic factors on the age-related decrease in thymic secretion. J Immunol 122:2502–2507
Bellamy D (1967) Hormonal effects in relation to ageing in mammals. Symp Soc Exp Biol 21:427–454
Berrih S, Arewana-Scisdedos F, Cohen S, Devos R, Charron D, Virelizier JL (1985) Interferon-gamma modulates HLA class II antigen expression on cultured human thymic epithelial cells. J Immunol 135:1165–1171
Bill J, Palmer E (1989) Positive selection of CD4+T cells mediated by MHC class II-bearing stromal cell in the thymic cortex. Nature 341:649–651
Bilder G (1976) Effect of endocrine manipulation on graft rejection. Immunol Commun 5:163–179
Calzolari A (1898) Recherches experimentales sur un rapport probable entre la function du thymus et celles des testicles. Arch Ital Biol 30:71–77
Castro JE (1974a) Orchidectomy and the immune response. I. Effect of orchidectomy on the lymphoid tissues of mice. Proc R Soc Lond [Biol] 185:425–436
Castro JE (1974b) Orchidectomy and the immune response. II. Respense of orchidectomised mice to antigens. Proc R Soc Lond [Biol] 185:437–451
Chiodi H (1940) The relationship between the thymus and the sexual organs. Endocrinology 26:107–116
Comsa J (1973) Hormonal interactions in the thymus. In: Luckey TD (ed) Thymic hormones. University Park Press, Baltimore
Comsa J, Leonhardt H, Ozminski K (1979) Hormonal influences on the secretion of the thymus. Thymus 1:81–93
Dallman MJ, Thomas ML, Green JR (1984) MRC Ox-19: a monoclonal antibody that labels rat T lymphocytes and augments in vitro proliferative responses. Eur J Immunol 14:260–267
Fabris N, Mocchegiani E (1985) Endocrine control of thymic serum factor production in young-adult and old mice. Cell Immunol 91:325–335
Farr AG, Sidman CL (1984) Reduced expression of Ia antigens by thymic epithelial cells of aged mice. J Immunol 133:98–103
Fitzpatrick FTA, Greenstein BD (1987) Effects of various steroids on thymic, spleen, prostate and seminal vesicles in old orchidectomised rats. J Endocrinol 113:51–55
Fitzpatrick FTA, Kendall MD, Wheeler MJ, Adcock IM, Greenstein BD (1985) Reappearance of the thymus of agein rats after orchidectomy. J Endocrinol 106:R17–19
Fitzpatrick FTA, Kendall MD, Wheeler MJ, Adcock IM, Greenstein BD (1986) Reappearance of thymus in ageing rats treated for one month with a stable analogue of luteinizing hormonereleasing hormone. J Endocrinol 108 [Suppl] Abstract 32
Fukumoto T, McMaster WR, Williams AF (1982) Mouse monoclonal antibodies against rat major histocompatibility antigens. Two Ia antigens and expression of Ia and class I antgens in rat thymus. Eur J Immunol 12:237–243
Furr BJA, Hutchinson FG (1985) Biodegradeable sustained-release formulation of the LH-RH analogue “Zoladex” for the treatment of hormone-responsive tumours. Proc Clin Biol Res 185A:143–153
Gordon H, Sweets HH (1936) A simple method for the silver impregnation of reticulum. Am J Pathol 12:545–552
Graff RS, Lappe MA, Snell GD (1969) The influence of the gonads and the adrenal glands on the immune response to skin grafts. Transplantation 7:105–111
Greenstein BD, Fitzpatrick FTA, Adcock IM, Kendall MD, Wheeler MJ (1986) Reappearance of the thymus in old rats after orchidectomy: inhibition of regeneration by testosterone. J Endocrinol 110:417–422
Greenstein BD, Fitzpatrick FTA, Kendall MD, Wheeler MJ (1987) Regeneration of the thymus in old male rats treated with a stable analogue of LHRH. J Endocrinol 112:345–350
Grossman CJ (1985) Interactions between the gonadal steroids and the immune system. Science 227:257–361
Hammar JA (1926) deÜber Methoden die Grösse und Anzahl der Hassallschen Körper des menschliche Thymus zahlenmässig festzustellen nebst einigen Worten über das numerische ‘Korpuskelproblem’ überhaupt. Z Mikrosk Anat Forsch 6:399
Henderson J (1904) On the relationship of the thymus to the sexual organs. I. The influence of castration on the thymus. J Physiol 31:22–229
Hirokawa K (1977) The thymus and aging. In: Makinodan T, Yunis EJ (eds) Immunology and Aging. Plenum Press, New York, pp 51–72
Hirokawa K, Makinodan T (1975) Thymic involution: effect on T cell differentiation. J Immunol 114:1659–1664
Iwata T, Incefy GS, Cunningham-Rundles C, Smithwick E, Geller N, O'Reilly R, Good RA (1981) Circulating thymic hormone activity in patients with primary and secondary immunodeficiency dicases. Am J Med 71:385–394
Jefferis WA, Green JR, Williams AF (1985) Authentic T helper CD4 (W3/25) antigen on rat peritoneal macrophages. J Exp Med 162:117–127
Johnson P, Gagnon J, Barclay AN, Williams AF (1985) Purification, chain separation and sequence of the MRC OX-8 antigen, a marker of rat cytotoxic T lymphocytes. EMBO (Eur Mol Biol Organ) J 4:2539–2545
Kay MMB (1978) Effect of age on T cell differentiation. Fed Proc 37:1241–1244
Kay MMB (1979) The thymus. Clock for immunological aging. J Invest Dermatol 73:29–38
Kay MMB, Makinodan T (1981) Relationship between aging and the immune system. Prog Allergy 29:134–181
Kieselow P, Boehmer H von (1990) Development of T cells in T-cell-receptor transgenic mice: direct evidence for positive and negative selection in the thymus. Thymus Update 3 (in press)
Korenchevsky VM, Dennison M, Shalit R (1932) The response of castrate water rats to the injection of testicular hormone. Biochem J 26:1306–1314
Kowalczyk D, Fournier M, Potworowski EF (1981) Age-dependent accumulation of thymic bormone-sensitive cells. Thymus 3:335–343
Lewis VM, Twomey JJ, Bealmear P, Goldstein G, Good RA (1978) Age, thymic involution and circulating thymic hormone activity. J Clin Endocr Metab 47:145–150
Makinodan T (1978) The thymus in ageing. In: Greenblatt RB. Geriatric Endocrinology, vol 5, Raven Press, New York, pp 217–230
Mason DY, Sammons R (1978) Alkaline phosphatase and peroxidase for double immunoenzymatic labelling of cellular constituents. J Clin Pathol 31:454–460
Oosterom R, Kater L (1981) The thymus in the ageing individual. II. Thymic epithelial function in vitro and in thymus pathology. Clin Immunol Pathol 18:195–202
Petricevic M, Denko CW, Messineo L (1981) Temporal changes of [3H] uridine and [14C] thymidine incorporation and total organ DNA and RNA in rat thymus. Thymus 2:355–359
Ready AR, Jenkinson EJ, Kingston R, Owen JJ (1984) Successful transplantation across major histocompatibility barrier of deoxygnanosine-treated embryonic thymus expressing class II antigens. Nature 310:231–233
Reem GH, Cook LA, Vilcek J (1983) Gamma interferon synthesis by human thymocytes and T lymphocytes inhibited by cyclosporin A. Science 221:63–65
Rock KL, Benacerraf B (1984) Thymic T cells are driven to expand upon interaction with self-class II major histocompatibility complex gene products on accessory cells. Proc Natl Acad Sci USA 81:1221–1224
Savino W, Dardenne M, Bach JF (1983) Thymic hormone containing cells. II. Evolution of cells containing the serum thymic factor (FTS or thymulin) in normal and autoimmune mice, as revealed by anti-FTS monoclonal antibodies. Relationship with Ia bearing cells. Clin Immunol 52:1–6
Schuurman H-J, van de Wijngaert FP, Delvoye L, Broekhuizen R, McClure JE, Goldstein AL (1985) Heterogeneity and age dependency of human thymus reticulo-epithelium in production of thymosin components. Thymus 7:13–23
Segre D, Segre M (1976) Humoral immunity in aged mice. II. Increased suppressor T cell activity in immunologically deficient old mice. J Immunol 116:735–738
Singhal SK, Roder JC, Duwe AK (1978) Suppressor cells in immunosenescence. Fed Proc 37:1245–1252
Singh J, Singh AK (1979) Age-related changes in the human thymus. Clin Exp Immunol 37:507–511
Smith CA, Williams GT, Kingston R, Jenkinson EJ, Owen JJT (1989) Antibodies to CD3/T-cell receptor complex induce cell death by apoptosis in immature T cells in thymic cultures. Nature 337:181–184
Steinmann GG, Müller-Hermelink H-K (1984) Lymphocyte differentiation and its microenvironment in the human thymus during aging. Monogr Dev Biol 17:142–155
Yunis EJ, Fernandez G, Good RA (1978) Ageing and involution of the immunological apparatus. In: Twomey JJ, Good RA (eds) The immunopathology of lymphoreticular neoplasms. Plenum Press, New York
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kendall, M.D., Fitzpatrick, F.T.A., Greenstein, B.D. et al. Reversal of ageing changes in the thymus of rats by chemical or surgical castration. Cell Tissue Res 261, 555–564 (1990). https://doi.org/10.1007/BF00313535
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00313535