Summary
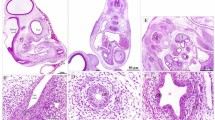
A light microscopical study of the morphogenesis of the gut in the rat embryo was undertaken to provide a careful map of temporal changes in the topographical relations of the (definitive) endoderm, the notochord and the hypoblast (primary endoderm). The borderline between the (definitive) endoderm and the hypoblast that appears upon gastrulation defines the lateral extension of the future gut epithelium. Within this initially semiglobular disk, the foregut and hindgut originate sequentially as blind, rapidly growing pouches. Upon the turning of the embryo, the hardly growing peripheral part of the disk becomes located in the vitelline duct. Within the head process, endodermal and notochordal cells could not be separately identified. However, slightly more posteriorly notochordal cells are seen to become embedded into the endoderm of the foregut during gastrulation. This process is not seen over the hindgut and may explain why the detachment of the notochord from the (fore) gut begins caudally.
Similar content being viewed by others
Abbreviations
- Arrow :
-
endoderm-hypoblast junction
- bar :
-
0.1 mm
- ect :
-
ectoderm
- end :
-
(definitive) endoderm
- fg :
-
foregut
- hg :
-
hindgut
- hp :
-
head process
- hyp :
-
hypoblast
- mes :
-
mesoderm
- not :
-
notochord
- ps :
-
primitive streak
- vd :
-
vitelline duct
References
Batten BE, Haar JL (1979) Fine structural differentiation of germ layers in the mouse at the time of mesoderm formation. Anat Rec 194:125–142
Beddington RSP (1982) An autoradiographic analysis of potency in different regions of the embryonic ectoderm during gastrulation in the mouse. J Embryol Exp Morphol 69:265–285
Beddington RSP (1983) The origin of foetal tissues during gastrulation in the rodent. In: Johnson MH (ed) Development in mammals. Elsevier, Amsterdam, vol 5, pp 1–32
Bellairs R (1953) Studies on the development of the foregut in the chick blastoderm. I. The presumptive foregut area. J Embryol Exp Morphol 1:115–124
Bellairs R (1986) The primitive streak. Anat Embryol 174:1–14
Fontaine J, Le Douarin NM (1977) Analysis of endoderm formation on the avian blastoderm by the use of quail-chick chimaeras. J Embryol Exp Morphol 41:209–222
Gardner RL, Rossant J (1979) Investigation of the fate of 4.5 day post coitum mouse inner cell mass cells by blastocyst injection. J Embryol Exp Morphol 52:141–152
Grobstein C (1952) Intra-ocular growth and differentiation of clusters of mouse embryonic shields cultured with and without primitive endoderm and in the presence of possible inductors. J Exp Zool 119:355–379
Hogan BLM, Newman R (1984) A scanning electron microscope study of the extra-embryonic endoderm of the 8th-day mouse embryo. Differentiation 26: 138–143
Jost A, Picon L (1970) Hormonal control of fetal development and metabolism. Adv Metab Disorders 4:123–184
Jurand A (1974) Some aspects of the development of the notochord in mouse embryos. J Embryol Exp Morphol 32:1–33
Lamers WH, Zonneveld D, Charles R (1984) Inducibility of carbamoyl-phosphate synthetase (ammonia) in cultures of embryonic hepatocytes: ontogenesis of the responsiveness to hormones. Dev Biol 105:500–508
Lawson KA, Meneses JJ, Pederson RA (1986) Cell fate and cell lineage in the endoderm of the presomite mouse embryo, studied with an intracellular tracer. Dev Biol 115:325–339
Levak-Svajger B, Svajger A (1974) Differentiation of endodermal tissues in homografts of primitive endoderm from two-layered rat embryonic shields. Experientia 27:683–684
Levak-Svajger B, Svajger A (1974) Investigation on the origin of the definitive endoderm in the rat embryo. J Embryol Exp Morphol 32:445–459
Nicolet G (1970) Avian gastrulation. Adv Morphogenesis 9:231–262
Poelmann RE (1981) The head-process and the formation of the definitive endoderm in the mouse embryo. Anat Embryol 162:41–49
Rosenquist GC (1966) A radioautographic study of labelled grafts in the chick endoderm. Contrib Embryol 38:71–110
Rosenquist GC (1972) Endoderm movements in the chick embryo between the early short streak and head process stage. J Exp Zool 180: 95–104
Schantz A, Schechter A (1965) Iron haematoxylin and safranins as polychrome stain for epon sections. Stain Technol 40:279
Schoenwolf GC, Franks MV (1984) Quantitative analyses of changes in cell shape during bending of the avian neural plate. Dev Biol 105:257–272
Schook P, Blom N (1978) A three-dimensional reconstruction method preserving light-microscopic and transmission electronmicroscopic information. Acta Morphol Neerl Scand 16:157–170
Schook P (1980) The effective osmotic pressure of the fixative for transmission and scanning electron-microscopy. Acta Morphol Neerl Scand 18:31–45
Snow MHL (1977) Gastrulation in the mouse: growth and regionalization of the epiblast. J Embryol Exp Morphol 42:293–303
Snow MHL (1981) Autonomous development of parts isolated from primitive-streak-stage mouse embryos. Is development clonal? J Embryol Exp Morphol [Suppl] 65:269–287
Svajger A, Levak-Svajger B (1974) Regional developmental capacities of the rat embryonic endoderm at the head-fold stage. J Embryol Exp Morphol 32:461–467
Tamarin A, Boyde A (1976) Three-dimensional anatomy of the 8-day mouse conceptus: a study by scanning electron microscopy. J Embryol Exp Morphol 36:575–596
Theiler K (1969) Die Anlage des Vorderdarmes bei der Hausmaus und die Furchenbildungen am Eizylinder. Z Entw Gesch 128:40–46
Theiler K (1972) The house mouse; development and normal stages from fertilization to 4 weeks of age. Springer, Berlin
Wiertz-Hoessels E, Drukker J (1987) Is there a specific interaction between donor embryonic entoderm (chick or quail) and host embryonic mesoderm (chick); submitted
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lamers, W.H., Spliet, W.G.M. & Langemeyer, R.A.T.M. The lining of the gut in the developing rat embryo. Anat Embryol 176, 259–265 (1987). https://doi.org/10.1007/BF00310060
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00310060