Summary
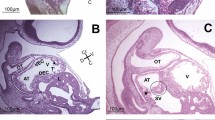
The development of the heart of Tupaia belangeri from the first endothelial-lined lumina to the cardiac loop is described in 20 embryos with 2 to 14 somites, from ontogenetic days 11 and 12. Bilateral endocardial tubes transporting blood are found in the 8-somite embryo; in the middle cardiac plate, angioblasts and angiocysts are located between them. In the 9-somite embryo, formation of the cardiac loop has started, the endocardial tubes approach each other closely, most of the angiocysts have been incorporated by the expanding endocardial tubes, and fusion of the endocardial lumina has started in the cono-truncal area. Apparently, much of the endocardial cardiac loop found in the 9-somite embryo has been produced by the disproportionate lengthening of a segment of the endocardial tubes, which is very short in the 8-somite embryo. In the 13-somite embryo the endocardial tubes have largely fused, but tube-like strands of endothelia, remnants of the original endothelial walls separating them, form a “palisade” and mark the original boundary between them. Myoepicardial differentiations of the splanchnopleure begin separately on both sides of the embryo and gradually spread craniad until they coalesce in the midline, in front of the anterior intestinal portal. The caudal portions of the endocardial tubes with initial myoepicardial and cardiac jelly differentiations do not contribute to the definitive heart. The anterior intestinal portal is very broad in Tupaia. Contradictions in the literature as to the bilaterality of cardiac primordia of eutherian mammals are discussed. The hypothesis is developed that bilateral endocardial tubes and bilateral myoepicardial differentiations of the splanchnopleure develop in species with a large yolk-sac, relatively late closure of the foregut, and a broad anterior intestinal portal (e.g., Tupaia, ferret, and cat, etc.). This is probably the primitive condition in eutherian mammals. In species with a small yolk-sac and/or reversal of germ layers (man, rodents), the foregut and anterior intestinal portal are formed earlier, the heart primordium reaches its median position ventral to the foregut in the angiocyst-stage, and the first endocardial lumina appear close to the midline. In these species, the primordium of the endocardium seems to be plexiform and without clear evidence for bilaterality.
Similar content being viewed by others
References
Bellairs R (1953) Studies in the development of the foregut in the chick blastoderm. II. The morphogenetic movements. J Embryol Exp Morphol 1:369–385
Bonnet R (1889) Beiträge zur Embryologie der Wiederkäuer, gewonnen am Schafei. Arch Anat Entw, pp 1–106
Bonnet R (1901) Beiträge zur Embryologie des Hundes. Erste Fortsetzung. Anat Hefte 16:231–332
Bremer JL (1912) The development of the aorta and aortic arches in rabbits. Am J Anat 13:111–128
Burlingame PL, Long JA (1944) The development of the heart in the rat. Univ Calif Publ Zool 43:249–319
Davis CL (1927) Development of the human heart from its first appearance to the stage found in embryos of twenty paired somites. Carnegie Inst Washington Contrib Embryol 19:245–284
DeHaan RL (1965) Morphogenesis of the vertebrate heart. In: DeHaan RL, Ursprung H (eds) Organogenesis. Holt, Reinhardt and Winston, New York, pp 377–419
DeVries PA (1981) Evolution of precardiac and splanchnic mesoderm in relationship to the infundibulum and truncus. In: Pexieder T (ed) Perspectives in cardiovascular research. Raven, New York 5:31–48
DeVries PA, Saunders JB de CM (1962) Development of the ventricles and spiral outflow tract in the human heart. A contribution to the development of the human heart from age group IX to age group XV. Contrib Embryol 37:87–114
Girgis A (1930) The development of the heart in the rabbit. Proc Zool Soc (London), pp 755–782
Girgis A (1933) A further contribution of the development of the heart in the rabbit. Proc Zool Soc (London), pp 421–450
Goss CM (1938) The first contractions of the heart in rat embryos. Anat Rec 70:505–524
Goss CM (1952) Development of the median coordinated ventricle from the lateral hearts in rat embryos with three to six somites. Anat Rec 112:761–796
Hensen V (1875/1876) Beobachtungen über die Befruchtung und Entwicklung des Kaninchens und Meerschweinchens. Z Anat Entwgesch 1:213–273, 353–423
Kaufman MH, Navaratnam V (1981) Early differentiation of the heart in mouse embryos. J Anat 133:235–246
Kriesell W (1977) Das Ovar von Tupaia. Unpubl M D Thesis, University of Göttingen
Kuhn H-J, Schwaier A (1973) Implantation, early placentation, and the chronology of embryogenesis in Tupaia belangeri. Anat Embryol 142:315–340
Mall FP (1912) On the development of the human heart. Am J Anat 13:249–298
Mollier S (1906) Die erste Anlage des Herzens bei den Wirbeltieren. In: Hertwig O (ed) Handbuch der vergleichenden und experimentellen Entwicklungslehre der Wirbeltiere. Fischer, Jena 1:1020–1051
O'Rahilly R (1971) The timing and sequence of events in human cardiogenesis. Acta Anat 79:70–75
O'Rahilly R (1973) Developmental stages in human embryos. Part A: Embryos of the first three weeks (Stages 1 to 9). Carnegie Institution, Washington DC, Publication 631
Pantke G (1981) Die Entstehung des Herzlumens. Unpubl M D Thesis University of Göttingen
Parker KM (1915) The early development of the heart and anterior vessels in marsupials, with special reference to Perameles. Proc Zool Soc (London), pp 459–499
Patten BM (1968) The development of the heart. In: Gould SE (ed) Pathology of the heart and blood vessels, 3rd edn. Thomas, Springfield, pp 20–90
Robinson A (1902) The early stages of the development of the pericardium. J Anat Physiol 37:1–17
Schulte H von W (1916) The fusion of the cardiac anlages and the formation of the cardiac loop in the cat (Felis domestica). Am J Anat 20:45–72
Schwaier A (1973) Breeding tupaias (Tupaia belangeri) in captivity. Z Versuchstierk 15:255–271
Seidl W, Steding G (1978) Topogenesis of the anterior intestinal port. Microkinematographic investigations on chick embryos. Z Anat Entwgesch 155:37–45
Seidl W, Steding G (1979) Frühstadien der Herzentwicklung (Gallus domesticus). Verh Anat Ges 73:577–579
Stalsberg H, DeHaan RL (1968) Endodermal movement during foregut formation in the chick embryo. Develop Biol 18:198–215
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kuhn, HJ., Liebherr, G. The early development of the heart of Tupaia belangeri, with reference to other mammals. Anat Embryol 176, 53–63 (1987). https://doi.org/10.1007/BF00309752
Issue Date:
DOI: https://doi.org/10.1007/BF00309752