Abstract
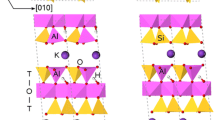
The electrostatic lattice energies of expanded and unexpanded micas are calculated starting from a “generic” structure the ionic charges of which are varied. The mode of expansion is to move the layers apart perpendicular to (001), the K+ ions remaining midway between the layers. The energy required for expansion is a quadratic function of the layer charge. It is larger when the layer charge is in the octahedral sites (K x Al2−x Mg x Si4O10(OH)2) than when it is in the tetrahedral sites (K x Mg3Si4−x Al x O10(OH)2).
Fluormicas have a slightly larger expansion energy than OH-micas. With the tetrahedral layer charge, dioctahedral micas have a slightly larger expansion energy than trioctahedral micas. This mode of expansion is less favourable than the mode usually adopted, viz. an expansion whereby the K ions divide themselves between the layers. The energy difference increases with the separation distance and is about 60 kJ mol−1 at 2.5 Å expansion. An intercalated water layer would be necessary to stabilize the K ions in positions midway between the layers.
Similar content being viewed by others
References
Appelo, C.A.J.: Layer deformation and crystal energy of micas and related minerals. I. Structural models for 1M and 2M 1 polytypes. Am. Mineral. 63, 782–792 (1978a)
Appelo, C.A.J.: Aspects of mica-related clay minerals in hydrogeochemistry. Thesis Vrije Universiteit, Amsterdam (1978b)
Bertaut, F.: L'énergie électrostatique de réseaux ioniques. J. Phys. Radium 13, 499–505 (1952)
Giese, R.F. Jr.: Surface energy calculations for muscovite. Nature (London) Phys. Sci. 248, 580–581 (1974)
Giese, R.F. Jr.: Interlayer bonding in talc and pyrophyllite. Clays Clay Miner. 23, 165–166 (1975a)
Giese, R.F. Jr.: The effect of F/OH substitution on some layersilicate minerals. Z. Kristallogr. 141, 138–144 (1975b)
Giese, R.F. Jr.: The influence of hydroxyl orientation, stacking sequence, and ionic substitutions on the interlayer bonding of micas. Clays Clay Miner. 25, 102–104 (1977)
Giese, R.F. Jr.: Hydroxyl orientations in 2:1 phyllosilicates. Clays Clay Miner. 27, 213–233 (1979)
Jenkins, H.D.B.: Truncation of Bertaut Madelung series for lattices involving extended charge distributions. Chem. Phys. Lett. 9, 473–475 (1971)
Jenkins, H.D.B.: Truncation of Bertaut Madelung series. Chem. Phys. Lett. 20, 155 (1973)
Jenkins, H.D.B. and Hartman, P.: A new approach to the calculation of electrostatic energy relations in minerals: the dioctahedral and trioctahedral phyllosilicates. Philos. Trans. R. Soc. London Ser. A: 293, 169–208 (1979)
Jenkins, H.D.B., Pratt, K.F.: Calculation of the first derivatives of Madelung constants with respect to cell lengths. Comput. Phys. Commun. 13, 341–348 (1978)
Jenkins, H.D.B., Pratt, K.F.: Convergence of the Bertaut series and the calculation of the electrostatic energy of an extended crystalline lattice. Chem. Phys. Lett. 62, 416–420 (1979)
Sidorenko, O.V., Zvyagin, B.B., Soboleva, S.V.: Crystal structure refinement for 1M dioctahedral mica. Kristallografiya 20, 543–549 (1975); Sov. Phys. Crystallogr. 20, 332–335 (1975)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Donald, H., Jenkins, B. & Hartman, P. Application of a new approach to the calculation of electrostatic energies of expanded Di- and trioctahedral micas. Phys Chem Minerals 6, 313–325 (1980). https://doi.org/10.1007/BF00307621
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00307621