Summary
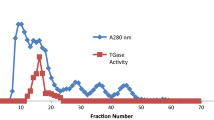
Available glutaraldehydes were fractionated on Sephadex G-10 in the pH range 3.0–8.2. Two fractions with absorptions at 235 and 280 mμ were separated, the latter alone having the aldehyde function. The relative elution volume of the aldehyde fraction was pH sensitive. Increase in temperature or pH increased the absorption at 280 mμ, the latter associated with a decrease in the aldehyde content. Glutaraldehyde solutions left three weeks at room temperature and pH 7.2 gave satisfactory fixation of rat liver.
Similar content being viewed by others
References
Andrews, P.: Estimation of the molecular weights of proteins by Sephadex Gel filtration. Biochem. J. 91, 222–233 (1964).
Blumenfeld, O. O., M. A. Paz, P. M. Gallop, and S. Seifter: The nature, quantity and mode of attachment of hexoses in ichthyocol. J. biol. Chem. 238, 3835–3839 (1963).
Caulfield, J. B.: Effects of varying the vehicle for OsO4 in tissue fixation. J. biochem. biophys. Cytol. 3, 827–830 (1957).
Coupland, R. E., and D. Hopwood: The mechanism of the differential staining reaction for Adrenaline and Noradrenaline storing granules in tissues fixed in Glutaraldehyde. J. Anat. (Lond.) 100, 227–243 (1966).
Fahimi, H. D., and P. Drochmans: Essais de standardisation de la fixation au glutaraldéhyde 1. Purification et détermination de la concentration du glutaraldéhyde. J. Microscopie 4, 725–736 (1965).
Fieser, L. F., and M. Fieser: Organic chemistry, 2nd ed. London: Harap 1958.
Flodin, P.: Dextran gels and their applications in gel filtration. Diss. 85 p. Uppsala, Sweden: AB Pharmacia 1962.
Gelotte, B.: Studies on gel filtration. Sorption properties of the bed material Sephadex. J. Chromatog. 3, 330–342 (1960).
Hopwood, D.: Some aspects of fixation with glutaraldehyde. A biochemical and histochemical comparison of the effects of formaldehyde and glutaraldehyde fixation on various enzymes and glycogen with a note on penetration of glutaraldehyde into rat liver. J. Anat. (Lond.) 101, 83–92 (1967).
Janigan, D. T.: Tissue enzyme fixation studies. 1. True effects of aldehyde fixation on β-glucuronidase, β-galactosidase, N-acetyl-β-glucosaminidase and β-glucosidase in tissue blocks. Lab. Invest. 13, 1038–1050 (1964).
: The effect of aldehyde fixation on acid phosphatase activity in tissue blocks. J. Histochem. Cytochem. 13, 473–483 (1965).
Kwon, T. W.: The pH dependant elution of malonaldehyde during gel filtration on Sephadex G-10. J. Chromatog. 24, 193–195 (1966).
, and B. M. Watts: Malonaldehyde in aqueous solution and its role as a measure of lipid oxidation in foods. J. Food Sci. 29, 294–302 (1964).
Maunsbach, A. B.: The influence of different fixatives and fixation methods on the ultrastructure of rat kidney proximal tubule cells II. Effects of varying osmolality, ionic strength, buffer system and fixative concentration of glutaraldehyde solutions. Ultrastruct. Res. 15, 283–309 (1960).
Sabatini, D. D., K. Bensch, and R. J. Barrnett: Cytochemistry and electron microscopy. The preservation of cellular ultrastructure and enzymatic activity by aldehyde fixation. J. Cell Biol. 17, 19–58 (1963).
Scott, A. L.: Interpretation of the ultraviolet spectra of natural products. Oxford: Pergamon Press 1964.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hopwood, D. The behaviour of various glutaraldehydes on Sephadex G-10 and some implications for fixation. Histochemie 11, 289–295 (1967). https://doi.org/10.1007/BF00305805
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00305805