Summary
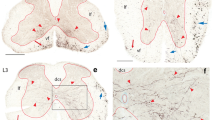
The synaptic relationships and the distribution of the afferent terminals of the spinal pathway to the lateral reticular nucleus (LRN) of the rat were examined following induced degeneration. After high cervical hemisections, the spino-LRN projection was first examined with the Fink-Heimer silver impregnation method. Degeneration was confined primarily to the ipsilateral LRN and all three divisions of the nucleus were involved. Maximum degeneration was observed in the caudal regions of the parvocellular division. The magnocellular division, except for the extreme dorsomedial area, showed substantial degeneration as well. The subtrigeminal division throughout its entire length contained only sparse degeneration.
Electron microscopic examination following spinal cord lesions revealed both round and pleomorphic-vesicle terminals in various stages of electron dense degeneration. The majority of the degenerating terminals were of the round-vesicle variety. Both types of terminals contacting somata were also observed to degenerate but their number was small in comparison to those on dendritic profiles. Terminals in synaptic contact with two dendritic profiles were also observed to degenerate. Some of the large terminals belonging to synaptic configurations (glomeruli) underwent degeneration and were therefore of spinal origin as well.
Similar content being viewed by others
References
Andrezik JA, King JS (1977) The lateral nucleus of the opossum (Didelphis virginiana). I. Conformation, cytology, and synaptology. J Comp Neurol 174:119–150
Brodal A (1949) Spinal afferents to the lateral reticular nucleus of the medulla oblongata in the cat. An experimental study. J Comp Neurol 91:259–295
Brodal P, Marsula J, Brodal A (1967) The cerebral cortical projection to the lateral reticular nucleus in the cat, with special reference to the sensorimotor cortical areas. Brain Res 6:252–274
Clendenin M, Ekerot C-F, Oscarsson O (1974a) The lateral reticular nucleus in the cat. III. Organization of component activated from ipsilateral forelimb tract. Exp Brain Res 21:501–513
Clendenin M, Ekerot C-F, Oscarsson O, Rosén I (1974b) Functional organization of two spinocerebellar paths relayed through the lateral reticular nucleus in the cat. Brain Res 69:140–143
Clendenin M, Ekerot C-F, Oscarsson O, Rosén I (1974c) The lateral reticular nucleus in the cat. II. Organization of component activated from bilateral ventral flexor reflex tract (bVFRT). Exp Brain Res 21:487–500
Clendenin M, Ekerot C-F, Oscarsson O (1975) The lateral reticular nucleus in the cat. IV. Activation from dorsal funiculus and trigeminal afferents. Exp Brain Res 24:131–144
Corvaja N, Grofová I, Pompeiano O, Walberg F (1977a) The lateral reticular nucleus in the cat. I. An experimental anatomical study of its spinal and supraspinal afferent connections. Neuroscience 2:537–553
Corvaja N, Grofová I, Pompeiano O, Walberg F (1977b) The lateral reticular nucleus in the cat. II. Effects of lateral reticular lesions on posture and reflex movements. Neuroscience 2:929–943
Courville J (1966) Rubrobulbar fibres to the facial nucleus and the lateral reticular nucleus (nucleus of the lateral funiculus). An experimental study in the cat with silver impregnation methods. Brain Res 1:317–337
Coulter JD, Mergner T, Pompeiano O (1974) Macular influences on ascending spinoreticular neurons located in the cervical cord. Brain Res 82:322–327
Coulter JD, Mergner T, Pompeiano O (1976) Effects of static tilt on cervical spinoreticular tract neurons. J Neurophysiol 39:45–62
Eccles JC (1964) The physiology of synapses. Springer-Verlag, New York, pp 201–215
Eccles JC (1973) The understanding of the brain. McGraw-Hill, New York
Ekerot C-F, Oscarsson O (1975) Inhibitory spinal paths to the lateral reticular nucleus. Brain Res 99:157–161
Fink RP, Heimer L (1967) Two methods for selective silver impregnation of degenerating axons and their synaptic endings in the central nervous system. Brain Res 4:369–374
Flumerfelt BA, Kapogianis EM, Hrycyshyn AW (1982) An ultrastructural study of the lateral reticular nucleus in the rat. Anat Embryol 165:329–344
Grillner S, Hongo T, Lunc S (1968) The origin of descending fibres monosynaptically activating spinorecticular neurons. Brain Res 10:259–262
Grundfest H, Carter WB (1954) Afferent relations of inferior olivary nucleus. I. Electrophysiological demonstration of dorsal spino-olivary tract in cat. J Neurophysiol 17:72–91
Hrycyshyn AW, Flumerfelt BA (1981a) A light microscopic investigation of the afferent connections of the lateral reticular nucleus in the cat. J Comp Neurol 197:477–502
Hrycyshyn AW, Flumerfelt BA (1981b) An electron microscopic study of the afferent connections of the lateral reticular nucleus of the cat. J Comp Neurol 197:503–516
Kapogianis EW, Flumerfelt BA, Hrycyshyn AW (1982) A Golgi study of the lateral reticular nucleus in the rat. Anat Embryol 164:243–256
Kawamura K, Brodal A, Hoddevik G (1974) The projection of the superior colliculus onto the reticular formation of the brain stem. An experimental anatomical study in the cat. Exp Brain Res 19:1–19
Kitai ST, DeFrance JF, Hatada K, Kennedy DT (1974a) Electrophysiological properties of lateral reticular nucleus cells. II. Synaptic activation. Exp Brain Res 21:419–432
Kitai ST, Kennedy DT, DeFrance JF, Hatada K (1974b) Electrophysiological properties of lateral reticular nucleus cells. I. Antidromic activation. Exp Brain Res 21:403–418
Kubin L, Magherini PC, Manzoni D, Pompeiano O (1980) Responses of lateral reticular neurons to sinusoidal stimulation of labyrinth receptors in decerebrate cat. J Neurophysiol 44:922–936
Künzle H (1973) The topographic organization of spinal afferents to the lateral reticular nucleus of the cat. J Comp Neurol 149:103–116
Ladpli R, Brodal A (1968) Experimental studies of commissural and reticular formation projections from the vestibular nuclei in the cat. Brain Res 8:65–69
Lundberg A, Oscarsson O (1962) Two ascending spinal pathways in the ventral part of the cord. Acta Physiol Scand 54:270–286
Martin GF, Andrezik J, Crutcher K, Linauts M, Panneton M (1977) The lateral reticular nucleus of the opossum. (Didelphis virginiana) II. Connections. J Comp Neurol 174:151–186
Mathers LH (1972) The synaptic organization of the cortical projection to the pulvinar of the squirrel monkey. J Comp Neurol 146:43–59
McLaughlin BJ (1972a) The fine structure of neurons and synapses in the motor nuclei of the cat spinal cord. J Comp Neurol 144:429–459
McLaughlin BJ (1972b) Propriospinal and supra-spinal projections to the motor nuclei in the cat spinal cord. J Comp Neurol 144:475–499
Mehler WR, Feferman ME, Nauta WJH (1960) Ascending axon degeneration following anterolateral cordotomy. An experimental study in the monkey. Brain 83:718–750
Mizuno N, Konishi A, Nakamura Y (1975) An electron microscope study of synaptic organization in the lateral reticular neucleus of the medulla oblongata in the cat. Brain Res 94:369–381
Mizuno N, Nakamura Y (1973) An electron microscope study of spinal afferents to the lateral reticular nucleus of the medulla oblongata in the cat. Brain Res 53:187–191
Morin F, Kennedy DT, Gardner E (1966) Spinal afferents to the lateral reticular nucleus. I. A histological study. J Comp Neurol 126:511–522
Nauta WJH (1957) Silver impregnation of degenerating axons. In: Windle WF (ed) New research techniques of neuroanatomy. Thomas, Springfield, pp 17–26
Oscarsson O (1967) Functional significance of information channels from the spinal cord to the cerebellum. In: Yahr MD, Purpura DP (eds) Neurophysiological basis of normal and abnormal motor activities. Raven/Hewlett, New York, pp 93–117
Oscarsson O (1973) Functional organization of spinocerebellar paths. In: Iggo A (ed) Handbook of sensory physiology. Vol. 2 Somatosensory systems. Springer-Verlag, New York, pp 339–380
Oscarsson O, Rosén I (1966) Response characteristics of reticulocerebellar neurons activated from spinal afferents. Exp Brain Res I:320–328
Rao GS, Breazile JE, Kitchell RL (1969) Distribution and termination of spinoreticular afferents in the brain stem of sheep. J Comp Neurol 137:185–196
Rosén I, Scheid P (1972) Cutaneous afferent responses in neurons of the lateral reticular nucleus. Brain Res 43:259–263
Rosén I, Scheid P (1973a) Patterns of afferent input in the lateral reticular nucleus of the cat. Exp Brain Res 18:242–255
Rosén I, Scheid P (1973b) Responses to nerve stimulation in the bilateral ventral flexor reflex tract (bVFRT) of the cat. Exp Brain Res 18:257–267
Rosén I, Scheid P (1973c) Responses in the spino-reticulo-cerebellar pathways to stimulation of cutaneous mechanoreceptors. Exp Brain Res 18:268–278
Szentágothai J (1970) Glomerular synapses, complex synaptic arrangements and their operational significance. In: Schmitt FO (ed) The neurosciences: second study program. Rockefeller Press, New York, pp 427–443
Uchizono K (1965) Characteristics of excitatory and inhibitory synapses in the central nervous system of the cat. Nature (Lond) 207:642–643
Verhaart WJC (1957) The lateral reticular nucleus of the medulla oblongata and the passing fibre systems of the lateral funiculus. Acta Psychiat Scand 32:211–229
Walberg F, Pompeiano O (1960) Fastigiofugal fibres to the lateral reticular nucleus: An experimental study in the cat. Exp Neurol 2:40–53
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Flumerfelt, B.A., Hrycyshyn, A.W. & Kapogianis, E.M. Spinal projections to the lateral reticular nucleus in the rat. Anat Embryol 165, 345–359 (1982). https://doi.org/10.1007/BF00305572
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00305572