Summary
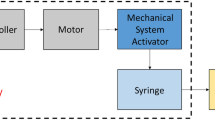
The tendency of insulin to form insoluble aggregates is a major obstacle to the development of implantable insulin infusion systems for treatment of insulin-deficient diabetic patients. A test system was developed to examine the kinetics of insulin aggregation under controlled conditions of temperature, vibration and contact material in an effort to provide design criteria for minimising aggregation. The contact materials tested were all potentially suitable for pump reservoirs on engineering criteria and included metals (stainless steel, titanium and a titanium alloy) and various plastics (polypropylene, polytetrafluoroethylene, Polyvinylchloride, polyamide, cellulose butyrate and silicone elastomer). The rate of insulin aggregation was markedly affected by the nature of the contact material. Hydrophilic materials, particularly polyamide and cellulose butyrate (2% of total insulin aggregated after 96 h vibration), appeared more compatible with insulin stability than did hydrophobic ones, such as polypropylene (16% aggregation) and Polyvinylchloride (37% aggregation). A specially formulated ‘pump’ insulin preparation, stabilised by addition of polyethylenepolypropyleneglycol, was significantly superior (three to five times more stable) to a regular neutral insulin formulation under most, but not all, conditions. Standard clinical syringes (polypropylene) performed poorly with both insulin formulations but especially with the neutral regular insulin (100% aggregation after 96 h vibration). In addition to physical aggregates, significant amounts (5%–30%) of the insulin remaining in solution were no longer detectable by immuno- or receptorassay in all materials tested. Appropriate combinations of insulin formulations and materials can minimise insulin aggregation and denaturation, but since the mechanisms involved are as yet poorly understood, realistic testing of proposed reservoir components and insulin formulations must be a prerequisite in insulin infusion pump planning and design. These testing procedures should be designed to test for denaturation in solution as well as for precipitation of insulin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Schade DS, Eaton RP, Edwards WS, Doberneck RC, Spencer WJ, Carlson GA, Bair RE, Love JT, Urenda RS, Gaona JI (1982) A remotely programmable insulin delivery system. JAMA 247: 1848–1853
Rupp WM, Barbosa JJ, Blackshear PJ, McCarthy HB, Rohde TD, Goldenberg FJ, Rublein TC, Dorman FD, Buchwald H (1982) The use of an implantable insulin pump in the treatment of type II diabetes. New Engl J Med 307: 265–270
Lougheed WD, Albisser AM (1980) Insulin delivery and the artificial beta cell: luminal obstructions in capillary conduits. Int J Art Org 3: 50–56
Lougheed WD, Woulfe-Flanagan H, Clement JR, Albisser AM (1980) Insulin aggregation in artificial delivery systems. Diabetologia 19: 1–9
Schade DS, Eaton RP, Delongo J, Saland LC, Ladman AJ, Carlson GA (1982) Electron microscopy of insulin precipitates. Diabetes Care 5: 25–30
Brownlee M, Cerami A, Li JJ, Vassara H, Martin TR, McAdam KPWJ (1984) Association of insulin pump therapy with raised serum amyloid A in type I diabetes mellitus. Lancet 1: 411–413
Albisser AM, McAdam KPWJ, Perlman K, Carson S, Bakoric A, Williamson JR (1983) Unanticipated amyloidosis in dogs infused with insulin. Diabetes 32: 1092–1101
Mauer SM, Buchwald H, Groppoli TJ, Rohde TD, Wigness BD, Rupp WM, Steffes MW (1983) Failure to find amyloidosis in dogs treated with long-term intravenous insulin delivered by a totally implantable pump. Diabetologia 25: 448–450
Lougheed WD, Albisser AM, Martindale HM, Chow JC, Clement JR (1983) Physical stability of insulin formulations. Diabetes 32: 424–432
Brange J, Havelund S (1983) Insulin pumps and insulin quality-requirements and problems. Acta Med Scand Suppl 671: 135–138
James DE, Jenkins AB, Kraegen EW, Chisholm DJ (1983) Insulin precipitation in artificial infusion devices. Diabetologia 21: 554–557
The United States Pharmacopeia (1980) Twentieth Revision, p 953
Peterson GL (1977) A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem 83: 364–356
Morgan CR, Lazarow A (1963) Immunoassay of insulin: two antibody system: plasma levels of normal, subdiabetic, diabetic rats. Diabetes 12: 115–126
Marshall RN, Underwood LE, Vorna SJ, Foushee DB, Van Wyk JJ (1974) Characterisation of the insulin and somatomedin-C receptors in human placental cell membranes. J Clin Endocrinol Metab 39: 283–292
Waugh DF (1948) Regeneration of insulin from insulin fibrils by the action of alkali. J Am Chem Soc 70: 1850–1857
Waugh DF (1950) Assay of insulin in vitro by fibril elongation and precipitation. J Biol Chem 185: 85–95
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Feingold, V., Jenkins, A.B. & Kraegen, E.W. Effect of contact material on vibration-induced insulin aggregation. Diabetologia 27, 373–378 (1984). https://doi.org/10.1007/BF00304853
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00304853