Summary
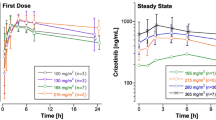
The plasma pharmacokinetic profile of 4′-epidoxorubicin (epirubicin) was investigated in 28 patients with nasopharyngeal carcinoma (NPC) after single i.v. rapid infusions. All patients had normal liver and renal functions. Plasma concentrations of the parent compound were specifically determined by a high-performance liquid chromatographic (HPLC) method, with UV detection at 254 nm. Plasma levels of the compound were fitted to a three-compartment open model; a triexponential decrease in plasma concentrations with a long terminal plasma halflife (44.8±21.2 h) was observed in 27 patients. The respective mean (±SD) serum concentration at 72 h and the AUC, plasma clearance, and terminal elimination rate constant in complete responders were 7.67±1.98 ng/ml, 4,002±3,080 ng· h/ml, 26.6±12.9 l/h·m2, and 0.009±0.007 l/h, whereas those in nonresponders were 4.96±1.8 ng/ml, 1,88±652.8 ng·h/ml, 44.4±15 l/h·m2, and 0.017±0.006 l/h, respectively; these differences were significant (P(0.05). Epirubicin produced a 52% response rate, including 6 patients with a complete response, 8 with a partial response, 11 with no change, and 2 with progressive disease. No relationship could be found between the various pharmacokinetic parameters and either leukopenia, age, or sex. These observations strongly suggest that plasma clearance may be one of the determining factors affecting the response or nonresponse of NPC patients to epirubicin, and a dose adjustment according to plasma clearance would probably increase the response rate.
Similar content being viewed by others
References
Arcamone F, Penco S, Vigevani A (1975) Adriamycin: new chemical developments and analogs. Cancer Chemother Rep 6: 123
Bonfante V, Bonadonna G, Villani F, Di Fronzo G, Martini A, Casazza AM (1979) Preliminary phase I study of 4′-epi-Adriamycin. Cancer Treat Rep 63: 915
Broggini M, Colombo T, Martini A, Donelli G (1980) Studies on the comparative distribution and biliary excretion of doxorubicin and 4′-epi-doxorubicin and mice and rats. Cancer Treat Rep 64: 897
Camaggi CM, Strocchi E, Tamassia V, Martoni A, Giovannini M, Iafelice G, Canova N, Marraro D, Martini A, Pannuti F (1982) Pharmacokinetic studies of 4′-epi-doxorubicin in cancer patients with normal and impaired renal function and with hepatic metastases. Cancer Treat Rep 66: 1819
Campora E, Teresa M, Seratoli MR (1984) Phase II study of 4′-epi-doxorubicin in advanced breast cancer. Cancer Treat Rep 68: 1285
Cancer Registry (1987) Annual report in the Taiwan area, 1984. Department of Health, Executive Yuan, Republic of China
Cersosimo RL, Hong WK (1986) Epirubicin: a review of the pharmacology, clinical activity, and adverse effects of an Adriamycin analogue. J Clin Oncol 4: 425
Chen KY (1984) Cancer registry biennial report 1982–1983. Veterans General Hospital, Taipei, Taiwan, Republic of China
Di Marco A, Casazza AM, Gambetta R, Supino R, Zunino F (1976) Relationship between activity and amino sugar stereochemistry of daunorubicin and Adriamycin derivatives. Cancer Res 36: 1962
Eksborg S, Stendahl U, Lonroth U (1986) Comparative pharmacokinetic study of Adriamycin and 4′-epi-Adriamycin after their simultaneous intravenous administration. Eur J Clin Pharmacol 30: 629
gibaldi M, Perrier D (1982) Pharmacokinetics, 2nd ed. Marcel Dekker, New York
Goodman TL, Loughner J (1986) Antibiotics in cancer chemotherapy: the anthracyclines. Hosp Formul Manage 21: 444
Hill BT, Whelan RDH (1982) A comparison of the lethal and kinetic effects of doxorubicin and 4′-epi-doxorubicin in vitro. Tumori 68: 29
Hurteloup P, Cappelaere P, Armand LP, Mathe G (1983) Phase II clinical evaluation of 4′-epi-doxorubicin. Cancer Treat Rep 67: 337
Kolaric K, Potrebica V, Cervek J (1983) Phase II clinical trial of 4′-epi-doxorubicin in metastatic solid tumors. J Cancer Res Clin Oncol 106: 148
Leyland-Jones B, Deesen P, Wittes R, Young CW (1982) High pressure liquid chromatography methodology and human plasma pharmacokinetics of 4′-epi-doxorubicin and its metabolites. Clin Pharmacol Ther February: 244
Libretti A (1984) Comparative clinical trial to investigate the safety and pharmacokinetics of 4′-epi-doxorubicin and doxorubicin. Confidential report of Farmitalia, IMI 28/606i
Maessen PA, Mross KB, Pinedo HM, Van Der Vijgh WJF (1987) Improved method for the determination of 4′-epi-doxorubicin and seven metabolites in plasma by high-performance liquid chromatography. J Chromatogr 417: 339
Metzler CM, Weiner DL (1986) PCNONLIN user's guide. Statistical Consultants, Lexington, Kentucky
Moro E, Bellotti V, Jannuzzo MG, Stegnjaich S, Valzella G (1983) High performance liquid chromatographic method for pharmacolinetic studies on the new anthracycline 4-demethoxy-daunorubicin and its 13-dihydro derivative. J Chromatogr 274: 281
Natale N, Brombilla M, Luchini S, Martini A, Moro E Pacciarini MA, Tamassia V, Vago G, Trabattono A (1981) 4′-Epi-doxorubicin and doxorubicin: toxicity and pharmacokinetics in cancer patients. Proceedings of the 12th International Congress of Chemotherapy, Florence, Italy, July, 19–24
Natale N, Piazza E, Italia C, Trabattoni A, Luchini S (1983) The kinetics of anthracycline in human plasma and tissues: daunorubicin, doxorubicin and 4′-epi-doxorubicin. Drugs Exp Clin Res 11: 775
Peterson C, Gunven P, Theve NO (1986) Comparative pharmacokinetics of doxorubicin in patients with gastrointestinal cancel. Cancer Treat Rep 70: 947
Robert J, Vrignaud P, Nguyen-Ngoc T Iliadis A, Mauriac L, Hurteloup P (1985) Comparative pharmacokinetics and metabolism of doxorubicin and epirubicin in patients with metastatic breast cancer. Cancer Treat Rep 59: 633
Sedman AJ, Wagner JG (1976) CSTRIP, a FORTRAN IV computer program for obtaining initial polyexponential parameter estimates. J Pharm Sci 65: 1006
Weenen H, Lankelma J, Penders PGM, McVie JG, Ten Bokkel Huinink WW, Planque MM de, Pinedo HM (1983) Pharmacokinetics of 4′-epi-doxorubicin in man. Invest New Drugs 1: 59
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hu, O.YP., Chang, SP., Jame, JM. et al. Pharmacokinetic and pharmacodynamic studies with 4′-epi-doxorubicin in nasopharyngeal carcinoma patients. Cancer Chemother. Pharmacol. 24, 332–337 (1989). https://doi.org/10.1007/BF00304769
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304769