Summary
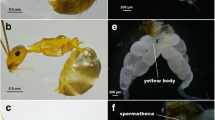
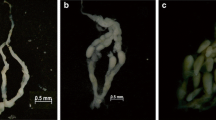
Two types of workers were recognized in colonies of Pristomyrmex pungens: extranidal workers (which characteristically walk outside the nest) and intranidal workers (which characteristically stay inside the nest). The ovaries of extranidal workers showed little activity, whereas those of intranidal workers showed high activity and often contained mature oocytes. I therefore conclude that only the intranidal workers reproduce. A behavioral repertoire of 103 individuals was obtained and used to infer group subdivision using cluster analysis; in addition, principal component analysis was performed on the intranidal workers in this set. These data enabled objective separation of extranidal and intranidal workers. Intranidal workers were larger in size on average than extranidal workers; however, the distributions overlapped. Three tests for further subdivision within the group of intranidal workers indicated that such subdivision is weak, and it is also likely that all intranidal workers lay eggs. There was no significant correlation between body size and reproductive status. The number of mature oocytes per ant fitted a Poisson distribution, and the first two principal component factors scores of behavior showed significant correlation with head width. All extranidal workers had resorbed ovaries and also had yellow bodies (which indicated a history of oviposition). When and how the differentiation between the reproductive intranidal workers and the non-reproductive extranidal workers occurred is discussed. The best-supported hypothesis is that extranidal workers are old intranidal ones. Neither males nor inseminated workers were found in any smaples collected in the field or studied in the laboratory, which greatly strengthens earlier suggestions that Pristomyrmex pungens is the first-known ant to be obligately thelytokous. These findings indicate that Pristomyrmex pungens is no longer eusocial, although it has the highest form of social behavior of any thelytokous species; they also raise the question of whether or not there are factors promoting the loss of eusociality and sexuality in this species. Ecological factors are tentatively indicated, namely, the need to maintain large colonies in the face of a nomadic lifestyle involving frequent colony fragmentation.
Similar content being viewed by others
References
Altmann J (1974) Observational study of behavior: sampling method. Behavior 49: 221–267
Brian MV (1974) Brood rearing behaviour in small culture of the ant Myrmica rubra L. Anim Behav 22: 879–889
Brian MV (1979) Caste differentiation and division of labor. In: hermann HR (ed) Social Insects, vol I. Academic Press, New York
Buschinger A (1986) Evolution of social parasitism in ants. Tree 1: 155–160
Cagniant H (1973) Apparition d'ouviéres chez la Fourmi Cataglyphis cursor Fonscolombe (Hyménoptéres, Formicidae). CR Acad Sci Ser D 277: 2197–2198
Cole BJ (1981) Dominance hierarchies in Leptothorax ants. Science 212: 83–84
Corbara B, Fresneau D, Lachaud J-P, Leelere Y, Goodall G (1986) An automated photographic technique for behavioural investigations of social insects. Behav Processes 13:237–249
Crozier RH (1975) Animal cytogenetics 3: Insecta (7) Hymenoptera. Gebrüder Bornträger, Berlin Stuttgart
Crozier RH (1982) On insects and insects: twists and turns in our understanding of evolution of sociality. In: Breed MD, Michener CD, Evans HE (eds) The biology of social insects. Westview Press, Boulder, pp 4–10
Franks NR, Scovell E (1983) Dominance and reproductive success among slave-making worker ants. Nature 304: 724–725
Fresneau D (1984) Development ovarien et statut social chez une fourmi primitive: Neoponera obscuricornis (Hym., Formicidac, Ponerinae). Insectes Soc 31: 387–402
Fresneau D, Garcia-Perez J, Jaisson P (1982) Evolution of polyethism in ants: observational results and theories. In: Jaisson P (ed) Social insects in the tropics, vol 1. Press de l'Université Paris-Nord, pp 129–155
Gadagkar R, Joshi NV (1983) Quantitative ethology of social wasps: Time-activity budgets and caste differentiation in Ropalidia marginata (Lep.) (Hymenoptera: Vespidae). Anim Behav 31: 26–31
Gadagkar R, Joshi NV (1984) Social organization in the Indian wasp Ropalidia cyatbiformis (Fab.) (Hymenoptera: Vespidae). Z Tierpsychol 64: 15–32
Hagan HR (1954) The reproductive system of the army-ant queen, Eciton (Eciton). Pt. I. General anatomy. Am Mus Novit 1663:1–12
Haskins CP, Whelden RM (1965) “Queenlessness”, worker sibship and colony vs population structure in Formicid genus Rhytidoponera. Psyche 72:87–112
Hölldobler B, Carlin NF (1985) Colony founding, queen dominance and oligogyny in the Australian meat ant Iridomyrmex purpureus. Behav Ecol Sociobiol 18: 45–58
Hölldobler B, Wilson EO (1977) The number of queens: an important trait in ant evolution. Naturwissenschaften 64:8–15
Itow T, Kobayashi K, Kubota M, Ogata K, Imai HT, Crozier RH (1984) The reproductive cycle of the queenless ant Pristomyrmex pungens. Insectes Soc 31:87–102
Mizutani A (1980) Preliminary report on worker oviposition in the ant Pristomyrmex pungens Mayr. Kontyû 48:327–332
Oster GF, Wilson EO (1978) Caste and ecology in the social insects. Princeton University Press, Princeton
Otto D (1958) Über die Arbeitsteilung in Staate von Formica rufa rufopratensis minor Gössw. und ihre verhaltensphysiologischen Grundlagen. Wiss Abh Dtsch Ackad Landwirtschaftswiss Berlin 30
Pamilo P, Crozier RH, Fraser J (1985) Inter-nest interactions, nest autonomy, and reproductive specialization in an Australian arid-zone ant, Rhytidoponera sp. 12. Psyche 92:217–236
Peeters CP (1987) The reproductive division of labour in the queenless ponerine ant Rhytidoponera sp. 12. Insectes Soc 34: 75–86
Peeters CP, Crewe R (1984) Insemination controls the reproductive division of labour in a ponerine ant. Naturwissenschaften 71:50–51
Peeters CP, Crewe R (1985) Worker reproduction in the ponerine and Ophthalmopone berthoudi: an alternative form of eusocial organization. Behav Ecol Sociobiol 18:29–37
Taylor RW (1965) The Australian ants of genus Pristomyrmex, with a case of apparent character displacement. Psyche 72:35–53
Teranishi C (1929) The habits and distributions of Japanese ants (II). Zool Mag 41:312–332 (in Japanese)
Tsuji K (1988) Nest relocation in the Japanese queeless ant, Pristomyrmex pungens Mayr (Hymenoptera: Formicidae). Insectes Soc (in press)
Ward JH (1963) Hierarchical grouping to optimize an objective function. J Am Stat Assoc 58:236–244
Ward PS (1983) Genetic relatedness and colony organization in a species complex of ponerine ants. I. phenotypic and genotypic composition of colonies. Behav Ecol Sociobiol 12:285–299
Weir JS (1958a) Polyethism in worker of the ant Myrmica, Pt I. Insectes Soc 5: 97–128
Weir JS (1958b) Polyethism in worker of the ant Myrmica, Pt II. Insectes Soc 5:315–359
Wheeler WM, Chapman JW (1922) The mating of Diacamma. Psyche 29:203–211
Wishart D (1969) An algolithm for hierarchical classifications. Biometrics 25:165–170
Wilson EO (1958) Studies on the ant fauna of Melanesia. I. The tribe Leptogenyni, II. The tribe Amblyoponini and Platythyrenini. Bull Mus Comp Zool Harv 118:101–153
Wilson EO (1971) The insects societeis. Harvard University Press, Cambridge
Wilson EO (1975) Sociobiology: the new synthesis. Belknap Press of Harvard University Press, Cambridge
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsuji, K. Obligate parthenogenesis and reproductive division of labor in the Japanese queenless ant Pristomyrmex pungens . Behav Ecol Sociobiol 23, 247–255 (1988). https://doi.org/10.1007/BF00302947
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00302947