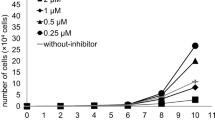
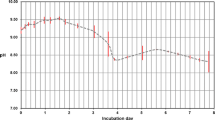
Summary
When embryonic cells in vitro are exposed to bromodeoxyuridine (BUdR) the duration of exposure can be made to last for several cell generation times. Such exposure is known to prevent embryonic cells undergoing terminal differentiation while leaving cell division and basic cell function unaffected. When BUdR is injected into pregnant mammals, it remains available for incorporation in the DNA for only a fraction of one S phase and causes foetal anomalies that are apparently the result of cell death and a transient slowing of the cell generation time but not of failure of cell lines to differentiate.
The objectives of our experiments were to ascertain the availability time of BUdR in the chicken embryo in ovo, to assess its teratogenicity and to examine its effects on the growth of the embryo.
When 3H-BUdR (0.02 mg) was injected into the albumen space on day 3 of incubation, subsequent scintillation spectrometry and autoradiography showed that the drug was incorporated into the DNA of the embryo for more than 8 h or more than one cell generation time at this stage of development. On the other hand, a trace amount of tritiated thymidine (3H-TdR) was available for only one hour, the difference being probably due to an expansion of the nucleotide precursor pool in the case of BUdR.
The injection of 0.02 mg BUdR on day 3 caused growth retardation as manifested by differences in weight and in DNA content between BUdR and saline treated embryos. The difference in DNA content was evident 24 h after treatment and was probably due in part of the cell necrosis in the developing CNS that began 10 h after injection. Differences in weight did not become apparent until 4 days after treatment and were thus thought to be due to factors other than cell necrosis.
On day 11 of incubation, the mortality of BUdR treated embryos became significantly greater than that of controls and many survivors after this time had ventral body wall defects. When treatment was delayed until days 4 or 6, the subsequent development of BUdR and saline treated embryos was indistinguishable. The sensitivity of day 3 was thought to be due to the fact that embryo DNA content quadrupled between days 3 and 4 whereas it only doubled per 24 h period thereafter.
Similar content being viewed by others
References
Agnish N, Kochar D (1976) Direct exposure of postimplantation mouse embryos to 5-bromodeoxyuridine in vitro and its effects on subsequent chondrogenesis in the limbs. J Embryol Exp Morphol 36:623–638
Atlas M, Bond VP (1965) The cell generation cycle of the eleven-day mouse embryo. J Cell Biol 26:19–24
Bannigan J, Langman J (1979) The cellular effect of 5-bromodeoxyuridine on the mammalian embryo. J Embryol Exp Morphol 50:123–135
Fabian B, Wilt F (1973) The incorporation of 5-bromodeoxyuridine into DNA of the area opaca vasculosa of the chick embryo. Dev Biol 32:92–100
Giles KW, Myers A (1965) An improved diphenylamine method for the estimation of deoxyribonucleic acid. Nature 206:93
Grabowski CT (1964) The etiology of hypoxia-induced malformations in the chick embryo. J Exp Zool 157:307–326
Grabowski CT (1966) Teratogenic effects of calcium salts on the chicken embryo. J Embryol Exp Morphol 15:113–118
Hamilton HL (1952) Lillie's development of the chick. In: Hamilton HL (ed) (Revised by H.L. Hamilton) Third ed. Henry Holt & Co, New York
Hilbelink D, Kaplan S (1978) Fast green staining of whole embryos for examination and photography. Stain Technol 53:261–264
Holtzer H, Weintraub H, Mayne R, Mochan B (1972) The cell cycle, cell lineage, and cell differentiation. In: Moscona AA Monray A (eds) Current topics in developmental Biology. Vol. 7. Academic Press, New York
Karnofsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J Cell Biol 27:137–138
Langman J, Haden C (1970) Formation and migration of neuroblasts in the spinal cord of the chick embryo. J Comp Neurol 138:419–432
Lee H, Despande A, Kalmus G (1974) Studies on the effects of 5-bromodeoxyuridine on the development of the explanted early chick embryo. J Embryol Exp Morphol 32:835–848
Munro H, Fleck A (1966) The determination of nucleic acids. In Glick D (ed) Methods of Biochemical Analysis. Vol. XIV. Interscience Publishers, New York-London-Sydney
Packard D, Menzies R, Skalko R (1973) Incorporation of thymidine and its analogue, bromodeoxyuridine, into embryos, and maternal tissues of the mouse. Differentiation 1:397–405
Palayoor T (1976) Transcriptional effects of 5-bromo-2-deoxyuridine in post-implantation mouse embryos. Experentia 33:448–450
Skalko R, Packard D, Schwendimann R, Raggio J (1971) The teratogenic response of mouse embryos to 5-bromodeoxyuridine. Teratology 4:87–94
Steck J, Nokata Y, Bader J (1969) The uptake of nucleosides by cells in culture. 1. Inhibition by heterologous nucleosides. Biochem Biophys Acta 190:237–249
Stellwagen R, Tomkins G (1971) Differential effect of 5-bromodeoxyuridine on the concentrations of specific enzymes in hepatoma cells in culture. Proc Natl Acad Sci USA 68:1417–1150
Wilt F, Anderson M (1972) The action of 5-bromodeoxyuridine on differentiation. Dev Biol 28:443–447
Xeros N (1962) Deoxyriboside control and synchronization of mitosis. Nature 194:682–683
Zagris N (1979) Differentiation capacity of unincubated chick blastoderm in culture. J Embryol Exp Morphol 50:47–55
Zamenhof S, Grauel L, Van Marthens E (1971) The effect of thymidine and 5-bromodeoxyuridine on developing chick embryo brain. Res Comm Chem Pathol Pharmacol 2:261–270
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bannigan, J., Langman, J. & van Breda, A. The uptake of 5-bromodeoxyuridine by the chicken embryo and its effects upon growth. Anat Embryol 162, 425–434 (1981). https://doi.org/10.1007/BF00301868
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00301868