Summary
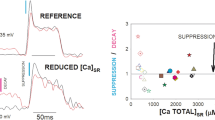
We have examined the spatial distribution of [Ca2+]i during tetanic stimulation in frog skeletal muscle cells using a fluorescence imaging method. We have found a completely unexpected pattern of Ca release: Ca is released forming gradients composed of spots of very significant and slow fluctuations of calcium release. Our findings could be explained if the calcium release process in skeletal muscle is influenced significantly by [Ca2+]i, such as in cardiac muscle, and suggests that the SR/Ca release control can include the established voltage-dependent plus a cardiac-like process of calcium-induced Ca release and a Ca release inhibition by Ca.
Similar content being viewed by others
References
BAYLOR, S. M., CHANDLER, W. K. & MARSHALL, M. W. (1979) Arsenazo II signals in singly dissociated frog twitch fibres. J. Physiol. 287, 23P-24P.
BAYLOR, S. M., CHANDLER, W. K. & MARSHALL, M. W. (1983) Sarcoplasmic reticulum calcium release in frog skeletal muscle fibres estimated from arsenazo III calcium transients. J. Physiol. 344, 625–66.
BERLIN, J. R., CANNELL, M. B. & LEDERER, W. J. (1989) Cellular origins of the transient inward current in cardiac myocytes. Role of fluctuations and waves of elevated intracellular calcium. Circ. Res. 65, 115–26.
Blinks, J. R. (1965) Influence of osmotic strength on cross-section and volume of isolated single muscle fibres. J. Physiol. 177, 42–57.
BREHM, P., LECHLEITER, J., SMITH, S. & DUNLAP, K. (1989) Intracellular signaling as visualized by endogenous calcium-dependent bioluminescence. Neuron 3, 191–8.
CANNELL, M. B. & ALLEN, D. G. (1984) Model of calcium movements during activation in the sarcomere of frog skeletal muscle. Biophys. J. 45, 913–25.
ELZINGA, G., HOWARTH, J. V., RALL, J. A., WILSON, M. G. A. & WOLEDGE, R. C. (1989) Variation in the normalized tetanic force of single frog muscle fibres. J. Physiol. 410, 157–70.
ENDO, M., TANAKA, M. & OGAWA, Y. (1970) Calcium induced release of calcium from the sarcoplasmic reticulum of skinned skeletal muscle fibers. Nature 228, 34–6.
HIROTA, A., CHANDLER, W. K., SOUTHWICK, P. L. & WAGGONER, A. S. (1989) Calcium signals recorded from two new purpurate indicators inside frog cut twitch fibers. J. Gen. Physiol. 94, 597–631.
HORIUTI, K., HIGUCHI, H., UMAZUME, Y., KONISHI, M., OKAZAKI, O. & KURIHAI, S. (1988) Mechanism of action of 2,3-butanedione 2-monoxime on contraction of frog skeletal muscle fibres. J. Muscle Res. Cell Motil. 9, 156–64.
Klein, M. G., Simon, B. J., Szucs, G. & Schneider, M. F. (1988) Simultaneous recording of calcium transients in skeletal muscle using high-and low-affinity calcium indicators. Biophys. J. 53, 971–88.
LECHLEITER, J., GIRARD, S., PERALTA, E. & CLAPHAM, D. (1991) Spiral calcium wave propagation and annihilation in Xenopus laevis oocytes. Science 252, 123–6.
LIPSCOMBE, D., MADISON, D. V., POENIE, M., REUTER, H., TSIEN, R. Y. & TSIEN, R. W. (1988) Spatial distribution of calcium channels and cytosolic calcium transient in growth cones and cell bodies of sympathetic neuron. Proc. Natl. Acad. Sci. 85, 2398–402.
MA, J., FILL, M., KNUDSON, C. M., CAMPBELL, K. P. & CORONADO, R. (1988) Ryanodine receptor of skeletal muscle is a gap junction-type channel. Science 242, 99–102.
MEISSNER, G. & HENDERSON, J. S. (1987) Rapid calcium release from cardiac sarcoplasmic reticulum vesicles is dependent on Ca2+ and is modulated by Mg2+ adenine nucleotide, and calmodulin. J. Biolog. Chem. 262, 3065–73.
MILEDI, R., PARKER, I. & ZHU, P. H. (1982) Calcium transients evoked by action potentials in frog twitch muscle fibres. J. Physiol. 333, 655–79.
MONCK, J. R., OBERHAUSER, A. F., KEATING, T. J. & FERNANDEZ, J. M. (1992) Thin-section ratiometric Ca2+ images obtained by optical sectioning of Fura-2 loaded mast cells. J. Cell Biol. 116, 745–59.
NEERING, I. R., QUESENBERRY, L. A., MORRIS, V. A. & TAYLOR, S. R. (1991) Nonuniform volume changes during muscle contraction. Biophys. J. 59, 926–32.
NIGGLI, E. & LEDERER, W. J. (1990a) Voltage-independent calcium release in heart muscle. Science 250, 565–8.
NIGGLI, E. & LEDERER, W. J. (1990b) Real-time confocal microscopy and calcium measurements in heart muscle cells towards the development of a fluorescence microscope with high temporal and spatial resolution. Cell Calcium 11, 121–30.
PAPE, P., KONISHI, M., HOLLINGWORTH, S. & BAYLOR, S. M. (1990) Perturbation of sarcoplasmic reticulum calcium release and phenol red absorbance transients by large concentration of Fura-2 injected into frog skeletal muscle fibers. J. Gen. Physiol. 96, 493–516.
PEACHY, L. D. & EISENBERG, B. R. (1978) Helicoids in the T system and striations of frog skeletal muscle fibers seen by high voltage electron microscope. Biophys. J. 22, 145–54.
ROUSSEAU, E. & PINKOS, J. (1990) pH modulates conducting and gating behaviour of single calcium release channels. Pflüger's Arch. 415, 645–7.
SCHNEIDER, M. F. & SIMON, B. J. (1988) Inactivation of calcium release from the sarcoplasmic reticulum in frog skeletal muscle. J. Physiol. 405, 727–45.
SHARNOFF, M., KARCHER, T. H. & BREHM, L. P. (1984) Microdifferential holography and the polysarcomeric unit of activation of skeletal muscle. Science 223, 822–5.
SOMLYO, A. V., GONZALEZ-SERRATOS, H., SHUMAN, H., McClellan, G. & Somlyo, A. P. (1981) Calcium release and ionic changes in the sarcoplasmic reticulum of tetanized muscle: An electron-probe study. J. Cell Biol. 90, 577–94.
TANABE, T., BEAM, K. G., ADAMS, B. A., NIIDOME, T. & NUMA, S. (1990) Regions of the skeletal muscle dihydropyridine receptor critical for excitation-contraction coupling. Nature 346, 567–9.
VAN DERKLOOT, W. G. (1969) The steps between depolarization and the increase in the respiration of frog skeletal muscle. J. Physiol. 204, 551–69.
VERGARA, J., DIFRANCO, M., COMPAGNON, D. & SUAREZ-ISLA, B. A. (1991) Imaging of calcium transients in skeletal muscle fibers. Biophys. J. 59, 12–24.
WESTERBLAD, H., LEE, J. A., LAMB, A. G., BOLSOVER, S. R. & ALLEN, D. G. (1990) Spatial gradients of intracellular calcium in skeletal muscle during fatigue. Pflüger's Arch. 415, 734–40.
WROGEMANN, K. & PENA, S. D. J. (1976) Mitochondrial calcium overload: a general mechanism for cell necrosis in muscle diseases. Lancet 27, 672–3.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rozycka, M., Gonzalez-Serratos, H. & Goldman, W. Non-homogeneous Ca release in isolated frog skeletal muscle fibres. J Muscle Res Cell Motil 14, 527–532 (1993). https://doi.org/10.1007/BF00297215
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00297215