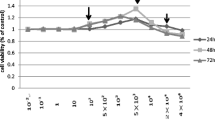
Summary
The skeleton is the major reservoir of lead and calcium in humans, and plays an important role in systemic calcium regulation. Lead perturbs normal calcium transport and second messenger function, directly or indirectly, in virtually all cells studies so far. Therefore, we and others have postulated that an early and discrete toxic effect of lead is perturbation of one or more loci within the calcium messenger system. To understand further the role of lead on calcium homeostasis in bone, we undertook this study to characterize calcium homeostasis and the effect of lead on calcium homeostasis in rat osteosarcoma (ROS 17/2.8) cells, which exhibit the osteoblast phenotype. ROS cells were incubated in medium containing 45Ca for 20 hours. Monitoring the efflux of 45Ca from the cultures for 210 minutes allowed for the determination of kinetic parameters defining steady state calcium homeostasis. Three distinct intracellular kinetic calcium pools characterized 45Ca homeostasis. Treatment with either 400 ng parathyroid hormone (PTH)/ml culture medium for 1 hour or 25 μM lead for 20 hours increased total cell calcium. Treatment with PTH caused a larger increase of cell calcium in lead-intoxicated cells than either lead intoxication or PTH treatment alone. This increase suggests that lead may perturb normal calcium-mediated PTH responsiveness of the osteoblast. These experiments further establish a kinetic model for the study of calcium homeostasis in osteoblastic bone cells. The studies also advance the hypothesis that lead-induced perturbations of calcium-mediated processes represent and early effect of lead toxicity at the cellular level.
Similar content being viewed by others
References
Agency for Toxic Substances and Disease Registry (1988) The nature and extent of lead poisoning in children in the United States. A report to Congress
Pounds JG, Long GJ, Rosen JF (1991) Cellular and molecular toxicity of lead in bone. Environ Health Perspect 91:17–32
Rabinowitz MB (1977) Wetherill GW, Kopple JD (1976) Kinetic analysis of lead metabolism in healthy humans. J Clin Invest 58:260–270
Needleman HL, Gunnoe C, Leviton A, Ree R, Peresic H, Maher C, Barrett P (1979) Deficits in psychologic and classroom performance of children with elevated dentine lead levels. N Engl J Med 300:689–696
Needleman HL, Schell A, Bellinger D, Leviton A, Allred EN (1990) The long-term effects of exposure to low doses of lead in childhood. An 11-year follow-up report. N Engl J Med 322:83–88
Rosen JF (1987) The toxicological importance of lead in bone: the evolution and potential uses of bone lead measurements by X-ray fluorescence to evaluate treatment outcomes in moderately lead toxic children. In: Clarkson TW (ed) The scientific basis and practical applications of biological monitoring of toxic metals. Plenum Press, NY, pp 603–621
Rosen JF, Markowitz ME, Bijur PE, Jenks ST, Wielopolski L, Kalef-Ezra JA, Slatkin DN (1989) L-line x-ray fluorescence of cortical bone lead compared with the CaNa2EDTA test in leadtoxic children: public health implications. Proc Natl Acad Sci 86:685–689
Somervialle LJ, Chettle DR, Scott MC, Tennant DR, McKierman MJ, Skilbeck A, Trethowan WN (1988) In vivo tibia lead measurements as an index of cumulative exposure in occupationally exposed subjects. Br J Indust Med 45: 174–181
Silbergeld EK, Schwartz J, Mahaffey K (1988) Lead and osteoporosis mobilization of lead from bone in postmenopausal women. Environ Res 47:79–94
Keller CA, Doherty RA (1980) Lead and calcium distributions in blood, plasma, and milk of the lactating mouse. J Lab Clin Med 95:81–89
Keller CA, Doherty RA (1980) Bone lead mobilization in lactating mice and lead transfer to suckling offspring. Toxicol Appl Pharmacol 55:220–228
Schwartz J, Angle CR, Pirkle JL, Pitcher H (1986) Childhood blood-lead levels and stature. Pediatrics 77:281–288
Shukla R, Bornschein RL, Dietrich KN, Mitchell T, Grote J, Berger O, Hammond PB, Succop PA (1987) Effects of fetal and early postnatal lead exposure on child's growth and stature—the Cincinatti lead study. In: Lindber SE and Hugchison TC (eds) Heavy metals in the environment, vol 1. CEP Consultants, Edinburgh, United Kingdom, pp 210–212
Long GJ, Rosen JF, Pounds JG (1990) Lead impairs the production of osteoclacin by rat osteosarcoma (ROS 17/2.8) cells. Toxicol Appl Pharmacol 106:270–277
Pounds JG (1984) Effect of lead intoxication on calcium homeostasis and calcium-mediated cell function: a review. Neurotoxicology 5:295–332
Simons TJB (1986) Cellular interactions between lead and calcium. Br Med Bull 42:431–434
Rosen JF, Chesney RW, Hamstra A, Deluca HF, Mahaffey KR (1980) Reduction in 1,25-dihydroxyvitamin D in children with increased lead absorption. N Engl J Med 302:1128–1131
Alkon DL, Rasmussen H (1988) A spatial-temporal model of cell activation. Science 239:998–1004
Pounds JG, Rosen JF (1986) Cellular metabolism of lead: a kinetic analysis in cultured osteoclastic bone cells. Toxicol Appl Pharmacol 83:531–545
Rosen JF, Pounds JG (1988) The cellular metabolism of lead and calcium: a kinetic analysis in cultured osteoclastic bone cells. Contrib Nephrol 64:74–82
Long GJ, Rosen JF, Pounds JG (1990) Cellular lead toxicity and metabolism in primary and clonal osteoblastic bone cells. Toxicol Appl Pharmacol 102:346–361
Pounds JG, Wright R, Morrison DR, Casciano DA (1982) Effect of lead on calcium homeostasis in the isolated rat hepatocyte. Toxicol Appl Pharmacol 63:389–401
Pounds JG, Morrison D, Wright R, Casciano D, Shaddock J (1982) Effect of lead on calcium-mediated cell function in the isolated rat hepatocyte. Toxicol Appl Pharmacol 63:402–408
Schacterle GR, Pollack RL (1973) A simplified method for the quantitative assay of small amounts of protein. Anal Biochem 51:654–655
Borlé AB (1990) Measurement of calcium movement across membranes: kinetic analysis and conceptualization. In: Intracellular calcium regulation, Alan R. Liss, New York, pp 19–75
Yamaoka K, Nakagawa T, Uno J (1978) Application of Akaike's information criterion in the evaluation of linear pharmacokinetic equations. J Pharmacokinet 6:165–175
Dowd TL, Rosen JF, Gupta RK (1991) 31P NMR and saturation transfer studies of the effect of Pb2+ on cultured osteoblastic bone cells. J Biol Chem 265:20833–20838
Rosen JF, Pounds JG (1989) Quantitative interactions between Pb2+ and CA2+ homeostasis in cultured osteoclastic bone cells. Toxicol Appl Pharmacol 98:530–543
Reid IA, Civitelli R, Halstead LR, Avioli LV, Hruska KA (1987) Parathyroid hormone acutely elevated intracellular calcium in osteoblast-like cells. Am J Physiol 252:E45-E52
Yamaguchi DT, Hahn TJ, Iida-Klein A, Kleeman CR, Muallem S (1987) Parathyroid hormone-activated calcium channels in an osteoblast-like clonal osteosarcoma cell line. J Biol Chem 262:7711–7718
Herrmann-Erlee MPM, Van der Meer JM, Löwik CWGM, Van Leeuwen JPTM, Boonekamp PM (1988) Different roles for calcium and cyclic AMP in the action of PTH: studies in bone explants and isolated bone cells. Bone 9:93–100
Abou-Samra A, Jueppner H, Westerberg D, Potts JT, Segre GV (1989) Parathyroid hormone causes translocation of protein kinase-C from cytosol to membranes in rat osteosarcoma cells. Endocrinology 124:1107–1113
Messing RO, Sneade AB, Savidge B (1990) Protein kinase C participates in up-regulation of dihydropyridine-sensitive calcium channels by ethanol. J Neurochem 55:1383–1389
Markovac J, Goldstein GW (1988) Picomolar concentrations of lead stimulate protein kinase C. Nature 334:372–374
Schanne FAX, Dowd TL, Gupta RK, Rosen JF (1990) Effect of lead on parathyroid hormone-induced responses in rat osteoblastic osteosarcoma cells (ROS 17/2.8) using 19F NMR. Biochim Biophys Acta 1054:250–255
McSheehy PMJ, Chambers TJ (1986) Osteoblastic-like cells in the presence of parathyroid hormone release soluble factor that stimulates osteoclastic bone resorption. Endocrinology 119: 1654–1659
Anderson C, Path MRC, FCAP, Danylchuk KD (1977) The effect of chronic low level lead intoxication on the haversian remodeling system in dogs. Lab Invest 37:466–469
Schwartz J, Angle CR, Pirkle JL, Pitcher H (1986) Childhood blood-lead levels and stature. Pediatrics 77:281–288
Angle CR, Thomas DJ, Swanson SA (1990) Lead inhibits the basal and stimulated response of a rat osteoblast-like cell line ROS 17/2.8 to 1α,25-dihydroxyvitamin D3 and IGF-1. Toxicol Appl Pharmacol 103:281–287
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Long, G.J., Pounds, J.G. & Rosen, J.F. Lead intoxication alters basal and parathyroid hormone-regulated cellular calcium homeostasis in rat osteosarcoma (ROS 17/2.8) cells. Calcif Tissue Int 50, 451–458 (1992). https://doi.org/10.1007/BF00296777
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00296777