Summary
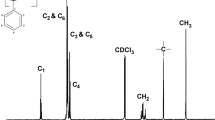
The title polymer was synthesized via free radical initiation. The 1H- and 13C-NMR spectra of the polymer were recorded and analyzed in terms of polymer tacticity. Many resonances showed a multiplicity indicative of sensitivity to stereochemistry. Those resonances that could be analyzed with a reasonable degree of accuracy showed poly(4-acetoxystyrene) to be an atactic polymer.
Similar content being viewed by others
References
D. L. Trumbo, T. Suzuki and H. J. Harwood, Polym. Bull., 4, 677 (1981).
D. L. Trumbo, Polym. Bull., 29, 377 (1992).
D. L. Trumbo, Polym. Bull., 33, 75 (1994), and references therein.
D. L. Trumbo, H. J. Harwood and T. K. Chen, Macromolecules, 14, 1138 (1981).
T. K. Chen, T. A. Gerkin and H. J. Harwood, Polym. Bull., 2, 37 (1980).
D. L. Trumbo and H. J. Harwood, Polym. Bull., 18, 27 (1987).
D. L. Trumbo and H. J. Harwood, Polym. Bull., 32, 207 (1993).
D. L. Trumbo, Polym. Bull., 33, 571 (1994).
D. L. Trumbo, Ph.D. Dissertation, The University of Akron, 1983.
K. J. Zhu, W. Liqun, W. Ji and Y. Shikn, Macromol. Chem. Phys., 195, 1965 (1994).
S. A. Heffner, M. E. Galuin, E. Reichmanis, L. Gerena and P. Mirau, ACS Symp. Ser., 614, 166 (1995).
R. Schwalm, D. Funhoff, H. Binder and H. Smuda, Eur. Pat. 605856 (1994).
J. M. Machado, U.S. Pat. 5210137 (1993).
L. M. Jackman and S. Sternhell, “Applications of Nuclear Magnetic Resonance Stereoscopy in Organic Chemistry”, Perhamon Press, New York (1972).
F. A. Bovey, “High Resolution NMR of Macromolecules”, Acad. Press, NY (1972).
In house computer program for calculating chemical shifts, S. C. Johnson Wax, Inc., Racine, WI (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Trumbo, D.L. Preliminary analysis of the 1H-NMR and 13C-NMR spectra of poly(4-acetoxystyrene). Polymer Bulletin 37, 617–623 (1996). https://doi.org/10.1007/BF00296607
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00296607