Summary
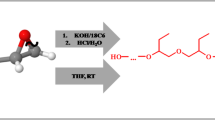
The synthesis of telechelic monodispersed diols produced from the radical telomerization of an excess of undecylenol with commercialy available α, ω-dithiols HSC2H4XC2H4SH (X=0, S or CH2) initiated by peroxides is presented. In each case, the diols were obtained selectively and quantitatively and they were characterized by both 1H and 13C NMR. Their physical characteristics (Tg, Tm and decomposition temperatures) were determined. Such compounds are thermally more stable than polydispersed telechelic commercially available diols.
Similar content being viewed by others
References
Ameduri B, Boutevin B (1990) Macromolecules
Ameduri B, Boutevin B (1991) Macromolecules 24:2475
Ameduri B, Berrada K, Boutevin B, Bowden RD submitted to Poly Bull
Harell LL (1969) Macromolecules 2:607
Bill R, Droschen M, Wegner G (1981) Makromol Chem 182:1033
Eisenbach CD, Gunter CL (1983) ACS Org Coat Appl Poly Sc Proc 49:239
Singh H, Williams M, Hutt JW, US Patent 0, 055, 531 (04/12/81)
Boutevin B, Pietrasanta Y (1990) in Allen G, Bevington JC (ed) Comprehensive Polymer Science, Pergamon Press, Vol III 14:185
Boutevin B, El Idrissi A, Parisi JP (1990) Makromol Chem 191:445
Boutevin B, Chaib M, Robin JJ (1990) Makromol Chem 191:737
Berrada K (1991), PhD thesis, Montpellier
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ameduri, B., Berrada, K., Boutevin, B. et al. Synthesis of telechelic monodispersed diols. Polymer Bulletin 28, 497–503 (1992). https://doi.org/10.1007/BF00296038
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00296038