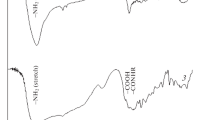
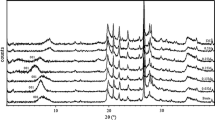
Abstract
The distribution of complexes of sodium picrate and potassium picrate with poly[(benzo-15-crown-5)maleimide], poly[(benzo-15-crown-5)maleimide-alt-α-methylstyrene] and benzo-15-crown-5 (b15/5) between water and CH2Cl2 was investigated. Concentration of Na-ion with homopolymer in CH2Cl2 was 52%, while the concentration of complex of alternating copolymer and b15/5 with Na-ion was 22% and 20% respectively. Concentration of K-ion complex in CH2Cl2 with homopolymer and alternating copolymer was 90% and with b15/5 the concentration of complex was 40%. The large difference in the complexation ability of Na-ion and K-ion with homopolymer and alternating copolymer is considered to be the consequence of the formation of 1:1 complex with Na-ion and 2:1 complex with K-ion.
Similar content being viewed by others
References
Hiraoka M (1982) Crown compounds, in Studies in Organic Chemistry 12, (Publ) Kodansha Ltd Tokuo and Elsevier Sci Publ Co, Amsterdam
Tunca U and Yagci Y (1994) Progr Polym Sci 19:233–286
Fleš DD, Hace D, Ranogajec F and Vuković R (1992) Polym Bull 29: 153
Pedersen C J (1967) J Am Chem Soc 89:7017
Kopolow S, Hogen-Esch TE and Smid J (1973) Macromolecules 6:133
Sadakane A, Iwachido T and Toei K (1975) Bull Chem Soc Japan 48:60
Pedersen CJ (1970) J Am Chem Soc 92:391
Ungaro R, ElHaj B and Smid J (1976) J Am Chem Soc 98:5198
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fles, D.D., Golja, G., Hace, D. et al. The extraction of sodium and potassium picrates with poly[(benzo-16-crown-5)maleimide], poly[(benzo-15-crown-5) maleimide-alt-α-methylstyrene] and benzo-15-crown-5. Polymer Bulletin 33, 425–430 (1994). https://doi.org/10.1007/BF00293486
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00293486