Summary
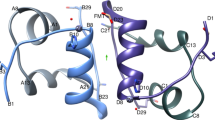
X-ray studies on semi-synthetic human insulin have shown that it crystallizes in the rhombohedral space group R3 and is nearly isomorphous with 2 Zn pig insulin. Precession photographs of crystals of human and pig insulins show observable changes in the intensity patterns. Crystallographic analysis and refinement of semi-synthetic human insulin at 1.9 Å resolution have shown that its molecular structure is very like that of pig insulin except at the C-terminus of the B chain where the change in sequence occurs. We also report the results of a high resolution crystallographic study of human insulins from different origins. The X-ray diffraction patterns of three non-pancreatic human insulins are indistinguishable from each other and from pancreatic human insulin. Refinement of the structures of the non-pancreatic human insulins has shown that they are identical within the limits of experimental error.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Markussen J, Damgaard U, Jorgensen KH, Snell L (1982) Production of human insulin from porcine insulin. In: Gueriguian JL, Bransone ED, Outschoorn AS (eds) Proceedings of the Joint FDA/USP Workshop on Drug and Reference Standards. USP Convention Inc, Rockville, Maryland, USA, pp 116–126
Schlichtkrull J (1958) Insulin crystals. Munksgaard, Copenhagen
Dodson EJ, Dodson GG, Hodgkin DC, Reynolds CD (1979) Structural relationships in the 2 Zn insulin hexamer. Can J Biochem 57: 469–479
North ACT, Phillips DC, Mathews FS (1968) A semi-empirical method of absorption correction. Acta Crystallographica A24: 351–359
Agarwal RC (1978) A new least-squares refinement technique based on the fast Fourier transform algorithm. Acta Crystallographica A34: 791–809
Baker EN, Dodson EJ (1980) Crystallographic refinement of the structure of actinidin at 1.7 Å resolution by fast Fourier leastsquares methods. Acta Crystallographica A36: 559–572
Sonnenberg GE, Chantelau E, Sundermann S, Hauff C, Berger M (1982) Human and porcine regular insulins are equally effective in subcutaneous replacement therapy. Diabetes 31: 600–602
Kemmer FW, Sonnenberg GE, Cuppers HJ, Berger M (1982) Absorption kinetics of semisynthetic human insulin and biosynthetic (recombinant DNA) human insulin. Diabetes Care 5 (Suppl 2):23–28
Clark AJL, Adeniyi-Jones RO, Knight G, Leiper JM, Wiles PG, Jones RH, Keen H, MacCuish AC, Ward JD, Watkins PJ, Cauldwell JM, Glynne A, Scotton JB (1982) Biosynthetic human insulin in the treatment of diabetes. Lancet 2: 354–357
Home PD, Aberti KGMM (1982) Human insulin. Clin Endocrinol Metab 11: 453–483
Blundell TL, Johnson LN (1976) In: Protein crystallography. Academic Press, New York, pp 404–419
Goeddel DV, Kleid DG, Bolivar F, Heyneker HL, Yansura DG, Crea R, Hirose T, Kraszewski A, Itakura K, Riggs AD (1979) Expression in E. coli of chemically synthesised genes for human insulin. Proc Natl Acad Sci USA 76: 106–110
Chance RE, Kroeff EP, Hoffman JA, Frank BH (1981) Chemical, physical and biological properties of biosynthetic human insulin. Diabetes Care 4: 147–154
Machin PA, Campbell JW, Elder M (eds) (1980) Refinement of protein structures. Proceedings of the Daresbury Study Weekend, SERC (Daresbury), Warrington, UK
Author information
Authors and Affiliations
Additional information
The determination of the human (semi-synthetic) 2 Zn insulin structure was carried out at the University of York. The part of the paper in which the human 2 Zn insulin crystal structures from different origins are compared was carried out at Birkbeck College (London) and the University of York.
Rights and permissions
About this article
Cite this article
Chawdhury, S.A., Dodson, E.J., Dodson, G.G. et al. The crystal structures of three non-pancreatic human insulins. Diabetologia 25, 460–464 (1983). https://doi.org/10.1007/BF00284451
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00284451