Abstract
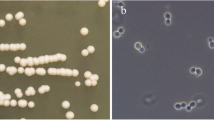
To obtain more information about the cell wall organization of Saccharomyces cerevisiae, we have developed a novel screening system to obtain cell wall-defective mutants, using a density gradient centrifugation method. Nine hypo-osmolarity-sensitive mutants were classified into two complementation groups, hpo1 and hpo2. Phase contrast microscopic observation showed that mutant cells bearing lesions at either locus became abnormally large. A gene that complemented the mutant phenotype of hpo2 was cloned and sequenced. This gene turned out to be identical to PKC1, which encodes the yeast homologue of mammalian protein kinase C. Complementation tests with pkc1Δ showed that hpo2 is allelic to pkc1. To study the reason for the fragility of hpo2 cells, cell wall was isolated and the glucan was analyzed. The amount of alkali, acid-insoluble glucan, which is responsible for the rigidity of the cell wall, was reduced to about 30% that of the wild-type cell and this may be the major cause of the fragility of the hpo2 mutant cell. Analysis of total wall proteins in hpo2 mutant cells on SDS-polyacrylamide gels revealed that a 33 kDa protein was overproduced two- to threefold relative to the wild-type level. This 33 kDa protein was identified as a β-glucanase, encoded by BGL2. Disruption of BGL2 in the hpo2 mutant partially rescued the growth rate defect. This suggests that the PKC1 kinase cascade regulates BGL2 expression negatively and overproduction of the β-glucanase is partially responsible for the growth defect. Since the bgl2 disruption did not rescue the hypo-osmolarty-sensitive phenotype of the hpo2 mutant, PKC1 must negatively regulate other enzymes involved in the biosynthesis and metabolism of the cell wall.
Similar content being viewed by others
References
Aldana CRV, Correa J, Segundo PS, Bueno A, Nebreda AR, Mendez E, Rey F (1991) Nucleotide sequence of the exo-1,3-β-glucanase-encoding gene, EXG1, of the yeast Saccharomyces cerevisiae. Gene 97:173–182
Ballou CE (1982) Yeast cell wall and cell surface. In: Strathern JN, Jones EW, Broach JR (eds) The molecular biology of the yeast Saccharomyces cerevisiae. Metabolism and gene expression. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, pp 335–358
Boone C, Sommer SS, Hensel A, Bussey H (1990) Yeast KRE genes provide evidence for a pathway of cell wall β-glucan assembly. J Cell Biol 110:1833–1843
Duffus JH, Levi C, Manners DJ (1982) Yeast cell-wall glucan. Adv Microb Physiol 23:151–181
Esmon PC, Esmon BE, Schauer IE, Tayor A, Schekman R (1987) Structure, assembly, and secretion of octameric invertase. J Biol Chem 262:4387–4394
Fleet GH (1991) Cell wall. In: Rose M (ed) The yeasts vol 4. Academic Press, New York, pp 199–278
Goud B, Salminen A, Walworth NC, Novic PJ (1988) A GTP-binding protein required for secretion rapidly associates with secretory vesicles and the plasma membrane in yeast. Cell 53:753–768
Hartwell LH (1967) Macromolecular synthesis in temperature-sensitive mutants of yeast. J Bacteriol 93:1662–1670
Hartwell LH, McLaughlin CS (1968) Temperature-sensitive mutants of yeast exhibiting a rapid inhibition of protein biosynthesis. J Bacteriol 96:1664–1671
Hien NH, Fleet GH (1983) Separation and characterization of six (1→3)-β-glucanases from Saccharomyces cerevisiae J Bacteriol 56:1204–1213
Hutchins K, Bussey H (1983) Cell wall receptor for yeast killer toxin: involvement of (1→6)-β-d-glucan. J Bacteriol 154:161–169
Katohda S, Abe N, Matsui M, Hayashibe M (1976) Polysaccharide composition of the cell wall of baker's yeast with special reference to cell walls obtained from large- and small-sized cells. Plant Cell Physiol 17:909–919
Klebl F, Tanner W (1989) Molecular cloning of a cell wall exo-β-1,3-glucanase from Saccharomyces cerevisiae. J Bacteriol 171:6259–6264
Lawrence CW (1991) Classical mutagenesis techniques. Meth Enzymol 194:273–280
Lee KS, Irie K, Gotoh Y, Watanabe Y, Araki H, Nishida E, Matsumoto K, Levin D (1993) A yeast mitogen-activated protein kinase homolog (Mpklp) mediates signalling by protein kinase C. Mol Cell Biol 13:3067–3075
Levin DE, Bartlett-Heubusch E (1992) Mutants in the S. cerevisiae PKC1 gene display a cell cycle-specific osmotic stability defect. J Cell Biol 116:1221–1229
Levin DE, Fields FO, Kunisawa R, Bishop JM, Thorner J (1990) A candidate protein kinase c gene, PKC1, is required for the S. cerevisiae cell cycle. Cell 62:213–224
Linnemans WAM, Boer P, Elbers PF (1977) Localization of acid phosphatase in Saccharomyces cerevisiae: a clue to cell wall formation. J Bacteriol 131:638–644
Manners DJ, Masson AL, Patterson JC (1973) The structure of a β-(1,3)-d-glucan from yeast cell wall. Biochem J 135:19–30
McMurrough I, Rose AH (1967) Effect of the growth rate and substrate imitation on the composition and structure of the cell wall of Saccharomyces cerevisiae. Biochem J 105:189–203
Mrša V, Klebl F, Tanner W (1993) Purification and characterization of the Saccharomyces cerevisiae BGL2 gene product, a cell wall endo-β-1,3-glucanase. J Bacteriol 175:2102–2106
Nebreda AR, Villa TG, Villanueva JR, Rey F (1986) Cloning of genes related to exo-β-glucanase production in Saccharomyces cerevisiae: characterization of an exo-β-glucanase structural gene. Gene 47:245–259
Oliver I, Hernandez LM, Larriba G (1985) Regulation of β-exoglucanase activity production by Saccharomyces cerevisiae in batch and continuous culture. Arch Microbiol 143:143–146
Paravicini G, Cooper M, Friedli L, Smith DJ, Carpentier JL, Klig LS, Payton MA (1992) The osmotic integrity of the yeast cell requires a functional PKC1 gene product. Mol Cell Biol 12:4896–4905
Pringle JR, Preston RA, Adams AM, Stearns T, Drubin DG, Haarer BK, Jones EW (1989) Fluorescence microscopy methods for yeast. Methods Cell Biol 31:357–435
Rey F, Villa TG, Santos T, Garcia-Acha I, Nombela C (1982) Purification and partial characterization of a new, sporulation specific, exo-β-glucanase from Saccharomyces cerevisiae. Biochem Biophys Res Commun 105:1347–1353
Roemer T, Bussey H (1991) Yeast β-glucan synthesis: KRE6 encodes a predicted type II membrane protein required for glucan synthesis in vivo and for glucan synthase activity in vitro. Proc Natl Acad Sci USA 88:11295–11299
Rose MD, Novick P, Thomas JH, Botstein D, Fink GR (1987) A Saccharomyces cerevisiae genomic plasmid bank based on centromere-containing shuttle vector. Gene 60:237–243
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Sherman F (1991) Getting started with yeast. Meth Enzymol 194:3–20
Sikorski RS, Hieter P (1989) A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 122:19–27
Stateva LI, Oliver SG, Trueman LJ, Venkov PV (1991) Cloning and characterization of a gene which determines osmotic stability in Saccharomyces cerevisiae. Mol Cell Biol 11:4235–4243
Yoshida S, Ikeda E, Uno I, Mitsuzawa H (1992) Characterization of staurosporine- and temperature-sensitive mutnt, sttl, of Saccharomyces cerevisiae: STT1 is allelic to PKC1. Mol Gen Genet 231:337–344
Author information
Authors and Affiliations
Additional information
Communicated by C.P. Hollenberg
Rights and permissions
About this article
Cite this article
Shimizu, J., Yoda, K. & Yamasaki, M. The hypo-osmolarity-sensitive phenotype of the Saccharomyces cerevisiae hpo2 mutant is due to a mutation in PKC1, which regulates expression of β-glucanase. Molec. Gen. Genet. 242, 641–648 (1994). https://doi.org/10.1007/BF00283417
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00283417