Summary
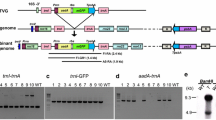
The tobacco (Nicotiana tabacum) nuclear genome contains long tracts of DNA (i.e. in excess of 18 kb) with high sequence homology to the tobacco plastid genome. Five lambda clones containing these nuclear DNA sequences encompass more than one-third of the tobacco plastid genome. The absolute size of these five integrants is unknown but potentially includes uninterrupted sequences that are as large as the plastid genome itself. An additional sequence was cloned consisting of both nuclear and plastid-derived DNA sequences. The nuclear component of the clone is part of a family of repeats, which are present in about 400 locations in the nuclear genome. The homologous sequences present in chromosomal DNA were very similar to those of the corresponding sequences in the plastid genome. However significant sequence divergence, including base substitutions, insertions and deletions of up to 41 bp, was observed between these nuclear sequences and the plastid genome. Associated with the larger deletions were sequence motifs suggesting that processes such as DNA replication slippage and excision of hairpin loops may have been involved in deletion formation.
Similar content being viewed by others
References
Albertini AM, Hofer M, Calos MP, Miller JH (1982) On the formation of spontaneous deletions: The importance of short sequence homologies in the generation of large deletions. Cell 29:319–328
Ayliffe MA, Timmis JN (1992) Tobacco nuclear DNA contains long tracts of homology to chloroplast DNA. Theor Appl Genet (in press)
Ayliffe MA, Timmis JN, Steele Scott N (1988) Homologies to chloroplast DNA in the nuclear DNA of a number of Chenopod species. Theor Appl Genet 75:282–285
Cheung WY, Steele Scott N (1989) A contiguous sequence in spinach nuclear DNA is homologous to three separated sequences in chloroplast DNA. Theor App] Genet 77:625–633
Dennis ES, Finnegan EJ, Taylor BH, Peterson TA, Walker AR, Peacock WJ (1988) Maize transposable elements: Structure, function and regulation. In: Nelson O (ed) Plant transposable elements. Plenum, New York
Drake JW, Glickman BW, Ripley LS (1983) Updating the theory of mutation. Am Sci 71:621–630
du Jardin P (1990) Homologies to plastid DNA in the nuclear and mitochondrial genomes of potato. Theor Appl Genet 79:807–812
Ellis J (1982) Promiscuous DNA-chloroplast genes inside plant mitochondria. Nature 299:678–679
Flavell RB (1988) DNA transposition — a major contributor to plant chromosome structure. BioEssays 1:21–22
Kamimura N, Ishii S, Liandong M, Shay JW (1989) Three separate mitochondrial sequences are contiguous in human genomic DNA. J Mol Biol 210:703–707
Li W-H, Luo C-C, Wu C-I (1985) Evolution of DNA sequences. In: MacIntyre RT (ed) Molecular evolutionary genetics
Moon E, Kae T-H, Wu R (1988) Rice mitochondrial genome contains a rearranged chloroplast gene cluster. Mol Gen Genet 213:247–253
Nags DK, Petes TD (1991) Even-base-pair inverted repeats in DNA form stable hairpins in vivo in Saccharomyces cerevisiae. Genetics 129:669–673
Ngernprasirtsiri J, Kobayashi H, Akazawa T (1988a) DNA methylation occurred around lowly expressed genes of plastid DNA during tomato fruit development. Plant Physiol 88:16–20
Ngernprasirtsiri J, Kobayashi H, Akazawa T (1988b) DNA methylation as a mechanism of transcriptional regulation in nonphotosynthetic plastid in plant cells. Proc Natl Acad Sci USA 85:4750–4754
Ngernprasirtsiri J, Chollet R, Kobayashi H, Sugiyama T, Akazawa T (1989) DNA methylation and the differential expression of C4 photosynthesis genes in mesophyll and bundle sheath cells of greening maize leaves. J Biol Chem 264:8241–8248
Nugent JM, Palmer JD (1991) RNA-mediated transfer of the gene coxII from the mitochondrion to the nucleus during flowering plant evolution. Cell 66:473–481
Ohme M, Kamogashira T, Shinozaki K, Sugiura M (1985) Structure and cotranscription of the tobacco chloroplast genes for tRNAGlu (UUC), tRNATyr (GUA) and tRNAAsp (GUC). Nucleic Acids Res 13:1045–1056
Ossorio PN, Sibley LD, Boothroyd JC (1991) Mitochondrial-like DNA sequences flanked by direct and inverted repeats in the nuclear genome of Toxiplasma gondii. J Mol Biol 22:525–536
Palmer JD, Thompson WF (1981) Clone banks of mung beank, pea and spinach chloroplast genomes. Gene 15:21–26
Pichersky E, Tanksley SD (1988) Chloroplast DNA sequences integrated into an intron of a tomato nuclear gene. Mol Gen Genet 215:65–68
Pichersky E, Longsdon JM (Jr), McGrath JM, Stasys RA (1991) Fragments of plastid DNA in the nuclear genome of tomato: prevalence, chromosomal location and possible mechanisms of integration. Mol Gen Genet 225:453–458
Ripley LS (1982) Model for the participation of quasi-palindrome DNA sequences in frameshift mutations. Proc Natl Acad Sci USA 79:4128–4132
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain termination inhibitors. Proc Natl Acad Sci USA 74:5463
Schaaper RM, Dansforth BN, Glickman BW (1986) Mechanisms of spontaneous mutagenesis: An analysis of the spectrum of spontaneous mutations in the Escherichia coli lacI gene. J Mol Biol 189:273–284
Schuster W, Brennicke A (1987) Plastid, nuclear and reverse transcriptase sequences in the mitochondrial genome of Oenothera: is genetic information transferred between organelles via RNA? EMBO J 6:2857–2863
Schuster W, Brennicke A (1988) Interorganellar sequence transfer: plant mitochondrial DNA is nuclear, is plastid, is mitochondrial. Plant Sci 54:1–10
Scott NS, Possingham JV (1980) Chloroplast DNA in expanding spinach leaves. J Exp Bot 123:1081–1092
Shimada H, Sugiura M (1991) Fine structural features of the chloroplast genome: Comparisons of sequenced chloroplast genomes. Nucleic Acids Res 19:983–995
Shinozaki K, Ohme M, Tanaka M, Wakasugi T, Hayashida N, Matsubayashi T, Zaita N, Chunwongse J, Obokata J, Yamaguchi-Shinozaki K, Ohto C, Torazawa K, Meng BY, Sugita M, Deno H, Kamogashira T, Yamada K, Kusuda J, Takaiwa F, Kato A, Tohdoh N, Shimada H, Sugiura M (1986) The complete sequence of the tobacco chloroplast genome: its organization and expression. EMBO 5:2043–2049
Steele Scott N, Timmis JN (1984) Homologies between nuclear and plastid DNA in spinach. Theor Appl Genet 67:279–288
Steele Scott N, Timmis JN, Ayliffe MA (1991) Chloroplast genes in higher plants. Aust J Plant Physiol 18:533–541
Stern DB, Palmer JD (1984) Extensive and widespread homologies between mitochondrial and chloroplast DNA in plants. Proc Natl Acad Sci USA 81:1946–1950
Sugiura M, Shinozaki K, Zaita N, Kusuda M, Kumano M (1986) Clone bank of the tobacco (Nicotiana tabacum) chloroplast genome as a set of overlapping restriction endonuclease fragments: mapping of eleven ribosomal protein genes. Plant Sci 44:211–216
Timmis JN, Steele Scott N (1983) Sequence homology between spinach nuclear and chloroplast genomes. Nature 305:65–67
Trinh TQ, Sinden RR (1991) Preferential DNA secondary structure mutagenesis in the lagging strand of replication of E. coli. Nature 352:544–547
van Loon LC, Trewavas A, Chapman KSR (1975) Phosphorylation of chromatin associated proteins in Lemna and Hordeum. Plant Physiol 55:288–292
Weaver DT, DePamphilis ML (1984) The role of palindromic and nonpalindromic sequences in arresting DNA synthesis in vitro and in vivo. J Mol Biol 180:961–986
Zurawski G, Perrot B, Bottomley W, Whitfeld PR (1981) The structure of the gene for the large subunit of the enzyme ribulose-1,5-bisphosphate carboxylase from spinach. Nucleic Acids Res 9:3251–3270
Author information
Authors and Affiliations
Additional information
Communicated by R. Hagemann
Rights and permissions
About this article
Cite this article
Ayliffe, M.A., Timmis, J.N. Plastid DNA sequence homologies in the tobacco nuclear genome. Molec. Gen. Genet. 236, 105–112 (1992). https://doi.org/10.1007/BF00279648
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00279648