Summary
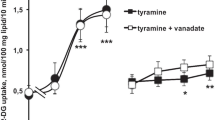
The effect of the catecholamine isoprenaline (10−5mol/l) and of the tumour promoting phorbolester tetradecanoyl-β-phorbol acetate (10−9mol/l) on insulin stimulated 3-O-methyl-glucose transport was studied in freshly isolated human adipocytes. Both substances reduced the maximal responsiveness of the glucose transport system to insulin by approximately 50%. To test if this is caused by inhibition of the insulin receptor kinase the receptor from phorbolester and isoprenaline treated cells was solubilized, partially purified and its kinase activity studied in vitro. Insulin stimulated 32P-incorporation into the β-subunit of the insulin receptor of phorbolester or isoprenaline treated cells was reduced to 20–60% of the values found with receptor from control cells at insulin concentrations between 10−10mol/l and 10−7mol/l. This inhibition of kinase activity of receptor from phorbolester and isoprenaline treated cells was observed at nonsaturating adenosine triphosphate levels (5 μmol/l), and it could be overcome with higher concentrations of γ-32P-adenosine triphosphate in the phosphorylation assay. A Lineweaver Burk analysis of the insulin stimulated receptor phosphorylation revealed that the Michaelis constant for adenosine triphosphate of the receptor kinase from phorbolester and isoprenaline treated cells was increased to >100 μmol/l compared with <50 μmol/l for receptor from control cells. We conclude from the data that catecholamine and phorbolester treatment of human adipocytes modulates the kinase activity of the insulin receptor by increasing its Michaelis constant for adenosine-triphosphate, and propose that this modulation of receptor kinase is a mechanism that can contribute to the pathogenesis of insulin resistance in human fat cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Rizza R, Haymond M, Cryer PE, Gerich J (1979) Differential effects of epinephrine on glucose production and disposal in man. Am J Physiol 237: E356-E362
Deibert C, DeFronzo RA (1980) Epinephrine-induced insulin resistance in man. J Clin Invest 65: 717–721
Chiasson JL, Shikama H, Chu DTW, Exton JH (1981) Inhibitory effect of epinephrine on insulin-stimulated glucose uptake by rat skeletal muscle. J Clin Invest 68: 706–713
Kirsch D, Baumgarten M, Deufel TH, Rinninger F, Kemmler W, Häring HU (1983) Catecholamine-induced insulin resistance of glucose transport in isolated rat adipocytes. Biochem J 216: 737–745
Kashiwagi A, Huecksteadt TP, Foley JE (1983) The regulation of glucose transport by cAMP stimulators via three different mechanisms in rat and human adipocytes. J Biol Chem 258: 13685–13692
Pessin JE, Gitomer W, Oka Y, Oppenheimer CL, Czech MP (1983) β-Adrenergic regulation of insulin and epidermal growth factor receptors in rat adipocytes. J Biol Chem 258: 7386–7394
Kirsch D, Obermaier B, Häring HU (1985) Phorbolesters enhance basal D-glucose transport but inhibit insulin stimulation of D-glucose transport and insulin binding in isolated rat adipocytes. Biochem Biophys Res Commun 128: 824–832
Häring HU, Kirsch D, Obermaier B, Ermel B, Machicao F (1986) Decreased tyrosine kinase activity of insulin receptor isolated from rat adipocytes rendered insulin-resistant by catecholamine treatment in vitro. Biochem J 234: 59–66
Häring HU, Kirsch D, Obermaier B, Ermel B, Machicao F (1986) Tumor-promoting phorbol esters increase the Km of the ATP-binding site of the insulin receptor kinase from rat adipocytes. J Biol Chem 261: 3869–3875
Kasuga M, Karlsson FA, Kahn CR (1982) Insulin stimulates the phosphorylation of the 95.000 Dalton subunit of its own receptor. Science 215: 185–187
Kasuga M, Zick Y, Blithe DL, Karlsson FA, Häring HU, and Kahn CR (1982) Insulin stimulation of phosphorylation of the β-subunit of the insulin receptor. J Biol Chem 257: 9891–9894
Häring HU, Kasuga M, and Kahn CR (1982) Insulin receptor phosphorylation in intact adipocytes and in a cell-free system. Biochem Biophys Res Commun 108: 1538–1545
Van Obberghen E, Kowalski A (1982) Phosphorylation of the hepatic insulin receptor. FEBS Lett 143: 179–192
Machicao F, Urumow T, Wieland OH (1982) Phosphorylation-de-phosphorylation of purified insulin receptor from human placenta. FEBS Lett 149: 96–100
Avruch J, Nemenoff RA, Blackshear PJ, Pierce MW, Osathanondh R (1982) Insulin stimulated tyrosine phosphorylation of the insulin receptor in detergent extracts of human placental membranes. J Biol Chem 257: 15162–15166
Petruzelli LM, Ganguly S, Smith CR, Cobbs MH, Rubin CS, Rosen O (1982) Insulin activates a tyrosine specific protein kinase in extracts of 3T3-L1 adipocytes and human placenta. Proc Natl Acad Sci USA 79: 6792–6796
Kasuga M, Zick Y, Blithe DL, Crettaz M, Kahn CR (1982) Insulin stimulates tyrosine phosphorylation of the insulin receptor in a “cell-free” system. Nature 298: 667–669
Zick Y, Kasuga M, Kahn CR, Roth J (1983) Characterization of insulin-mediated phosphorylation of the insulin receptor in a cell-free system. J Biol Chem 258: 75–80
Kasuga M, Fujita-Yamaguchi D, Blithe L, Kahn CR (1983) Tyro-sine-specific protein kinase activity is associated with the purified insulin receptor. Proc Natl Acad Sci USA 80: 2137–2141
Van Obberghen E, Rossie B, Kowalski A, Gazzano H, Ponzio G (1983) Receptor-mediated phosphorylation of the hepatic insulin receptor evidence that the M = 95.000 receptor subunit is its own kinase. Proc Natl Acad Sci USA 89: 945–949
Yu KT, Czech MP (1984) Tyrosine phosphorylation of the insulin receptor β-subunit activates the receptor-associated tyrosine kinase activity. J Biol Chem 259: 5277–5286
Rodbell M (1964) Metabolism of isolated fat cells. I. Effect of hormones on glucose metabolism and lipolysis. J Biol Chem 239: 375–380
Häring HU, Biermann E, Kemmler W (1981) Coupling of insulin binding and insulin action on glucose transport in fat cells. Am J Physiol 240: E 556-E 565
Obermaier B, Biemer E, Neffe J, Häring HU (1985) Structure and kinase activity of the insulin receptor in human fat and sceletal muscle. Diabetes 34 [Suppl 1]: 163A
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 227: 680–685
Häring HU, White MF, Kahn CR, Kasuga M, Lauris V, Fleischmann R, Murray M, Pawelek J (1984) Abnormality of insulin binding and receptor phosphorylation in an insulin resistant melanoma cell line. J Cell Biol 99: 900–908
Kadowaki T, Kasuga M, Akanuma Y, Ezaki O, Takaku F (1984) Decreased autophosphorylation of the insulin receptor kinase in streptozotocin-diabetic rats. J Biol Chem 259: 14208–14216
Häring HU, Kirsch D, Hölzl J, Herberg L, Eckel J (1984) Protein kinase activity of the insulin receptor from muscle of normal rats and of diabetic Zucker rats. Diabetologia 27: 283A
LeMarchand Y, Gremeauy T, Ballotti R (1985) Insulin receptor tyrosine kinase is defective in skeletal muscle of insulin-resistant obese mice. Nature 315: 676–679
Häring HU, Kasuga M, White MF, Crettaz M, Kahn CR (1984) Phosphorylation and dephosphorylation of the insulin receptor: evidence against an intrinsic phosphatase activity. Biochemistry 23: 3298–3306
Jacobs S, Sahoyoun NE, Saltiel AR, Cuatrecasas P (1983) Phorbol esters stimulate the phosphorylation of receptors for insulin and somatomedin. Proc Natl Acad Sci USA 80: 6211–6213
Takayama S, White MF, Lauris V, Kahn CR (1984) Phorbol esters modulate insulin receptor phosphorylation and insulin action in cultured hepatoma cells. Proc Natl Acad Sci USA 81: 7790–7801
Grigorescu F, Flier JS, Kahn CR (1984) Defect in insulin receptor phosphorylation in erythrocytes and fibroblasts associated with severe insulin resistance. J Biol Chem 259: 15003–15006
Grunberger G, Zick Y, Gordon PH (1984) Defect in phosphorylation of insulin receptors in cells from an insulin-resistant patient with normal insulin binding. Science 223: 932–934
Grunberger G (1984) Tyrosine kinase activity of insulin receptor of patients with type A extreme insulin resistance. Studies with circulating mononuclear cells and cultured lymphocytes. J Clin EndocrinolMetab 59: 5
Rovera G, O'Brien TG, Diamond L (1979) Induction of differentiation in human pyemyelocytic leukemia cells by tumor promoters. Science 204: 868–870
Dicker P, Rozengurt E (1980) Phorbolesters and vasopressin stimulate DNA syntheses by a common mechanism. Nature 287: 607–612
Rohrschneider LR, O'Brien DH, Boutwell RK (1972) Destimulation of phospholipide metabolism in mouse skin following phorbolester-treatment. Biochim Biophys Acta 280: 57–70
Castagna M, Takai Y, Kaibuchi K, Sano K, Kikkawa U, Nishizuka Y (1982) Direct activation of calcium-activated, phospholipid-dependent protein kinase by tumor promoting phorbol esters. J Biol Chem 257: 7847–7851
Nishizuka Y (1983) Phospholipid degradation and signal translation for protein phosphorylation. Trends Biochem Sci 8: 13–16
Kono T, Robinson FW, Sarver JA, Vega FV, Pointer RH (1977) Actions of insulin in fat cells. J Biol Chem 252: 2226–2233
White MF, Häring HU, Kasuga M, Kahn CR (1984) Kinetic properties and sites of autophosphorylation of the partially purified insulin receptor from hepatoma cells. J Biol Chem 259: 255–264
Stadtmauer L, Rosen OM (1986) Increasing cAMP content of IM-9 cells alter the phosphorylation state and protein kinase activity of insulin receptor. J Biol Chem 261: 3402–3407
Roth RA, Beandoin J (1986) Phosphorylation of purified insulin receptor by cAMP-kinase. Diabetes (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Obermaier, B., Ermel, B., Kirsch, D. et al. Catecholamines and tumour promoting phorbolesters inhibit insulin receptor kinase and induce insulin resistance in isolated human adipocytes. Diabetologia 30, 93–99 (1987). https://doi.org/10.1007/BF00274578
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00274578