Summary
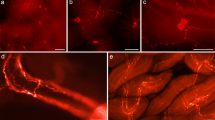
A tryptophanyl-tRNA synthetase (TrpRS)-immunoreactivity is localized in various neurosecretory cells of all ganglia of the central nervous system of the Orthoptera Locusta migratoria, except in deutocerebrum, and in endocrine cells of the midgut. It has been observed that TrpRS-like material never co-localizes either with CCK-like or octopamine-like material.
TrpRS immunoreactive perikarya and processes that ramify extensively throughout the neuropiles have been detected in the protocerebrum, optic lobes, tritocerebrum, suboesophageal, thoracic and abdominal ganglia. In the lateral protocerebrum, a particular TrpRS pathway different from the lateral gastrin cholecystokinin (CCK-8(s)) pathway is revealed, certain of these processes terminating in the glandular part of the corpora cardiaca. In the metathoracic ganglion, have been observed numerous immunoreactive cell bodies and processes in the neuropiles. Some of them constitute a major pathway and which are distinct from octopamine (OA) cells but in close vicinity with the latter. In the midgut immunopositive TrpRS-like cells are dispersed among the regenerative and digestive cells of the epithelium; they are different from gastrin-cholecystokinin positive cells.
The various TrpRS-like immunoreactivities identified in Locusta indicate that TrpRS-like material may occur in different tissues of organisms other than Vertebrates. These results suggest also that TrpRS-like enzyme could be involved in functions other than aminoacylation, as in Vertebrates.
Similar content being viewed by others
References
Andrews D, Trezeguet V, Merle M, Graves PV, Muench KH, Labouesse B (1985) Tryptophanamide formation by Escherichia coli tryptophanyl-tRNA synthetase. Eur J Biochem 146:201–209
Andries JC, Beauvillain JC (1988) Ultrastructural study of cholecystokinin-like immunoreactivity in endocrine cells of the insect midgut. Cell Tissue Res 254:75–81
Atkins R, Lambovitz AM (1982) A protein required for splicing group I introns in Neurospora mitochondria is mitochondrial Tyrosyl-tRNA synthetase as a derivative there of. Cell 50:331–345
Herbert CJ, Labouesse B, Dujardin G, Slonimski PP (1986) The NAM2 proteins from S. cerevisiae and S. douglasii are mitochondrial leucyl-tRNA synthetases and are involved in mRNA splicing. EMBO J 7:473–483
Hoyle G (1975) Evidence that insect dorsal unpaired median (DUM) neurons are octopaminergic. J Exp Zool 193:425–431
Hoyle G (1978) The dorsal, unpaired, median neurons of the locust metathoracic ganglion. J Neurobiol 9:43–57
Konings PNM, Vullings HGB, Geffard M, Buijs RM, Diederen JHB, Jansen WF (1988) Immunocytochemical demonstration of octopamine-immunoreactive cells in the nervous system of Locusta migratoria and Schistocerca gregaria. Cell Tissue Res 251:371–379
Lee CC, Craigen WJ, Muzny DM, Harlow E, Caskey CT (1990) Cloning and expression of a mammalian peptide chain release factor with sequence similarity to tryptophanyl-tRNA synthetase. Proc Natl Acad Sci USA 87:3508–3512
Mérault G, Graves PV, Labouesse B, Labouesse J (1978) Influence of magnesium on the steady-state derived order of substrate addition and product release in tRNATrp aminoacylation by beef pancreas tryptophan-tRNA ligase. Eur J Biochem 87:541–550
Mons N, Geffard M (1987) Specific antisera against the catecholamines: l-dihydroxyphenylalanine, dopamine, noradrenaline and octopamine tested by an enzyme-linked immunosorbent assay. J Neurochem 48:1826–1833
Nakane PK (1968) Simultaneous localization of multiple tissue antigens using the peroxidase-labelled antibody method: a study on pituitary glands of the rat. J Histochem Cytochem 16:557–560
Orchard J (1987) Adipokinetic hormones — an update. J Insect Physiol 33:451–463
Sallafranque ML, Garret M, Benedetto JP, Fournier M, Labouesse B, Bonnet J (1986) Tryptophanyl-tRNA synthetase is a major soluble protein species in bovine pancreas. Biochim Biophys Acta 882:192–199
Schooneveld H, Romberg-Privee HM, Veenstra JA (1986) Immunocytochemical differentiation between adipokinetic hormone (AKH)-like peptides in neurons and glandular cells in the corpus cardiacum of Locusta migratoria and Periplaneta americana with C-terminal and N-terminal specific antisera to AKH. Cell Tissue Res 243:9–14
Sternberger LA, Hardy JPH, Cuculis JJ, Meyer HG (1970) The unlabeled antibody method of immunohistochemistry: preparation and properties of soluble antigen-antibody complex (horseradish peroxidase-antihorseradish peroxidase) and its use in identification of spirochetes. J Histochem Cytochem 18:315–333
Sternberger LA (1986) Immunohistochemistry, 3rd edn. Wiley, New York
Tamarelle M, Romeuf M, Vanderhaeghen JJ (1988) Immunohistochemical localization of gastrin-cholecystokinin-like material in the central nervous system of the migratory locust. Histochemistry 89:201–207
Vaitukaitis JL (1981) Production of antisera with small doses of immunogen: multiple intradermal injections. Methods Enzymol 73:46–52
Vanderhaeghen JJ, Lotstra F, De Mey J, Gilles C (1980) Immunohistochemical localization of cholecystokinin and gastrin-like peptide in the brain and hypophysis of the rat. Proc Natl Acad Sci USA 77:1190–1194
Vandesande F (1988) Double and multiple immunoenzymatic labelling of tissue sections for light microscopy. Acta Histochem 35:107–115
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tamarelle, M., Sallafranque, M.L., Benedetto, J.P. et al. Tryptophanyl-tRNA synthetase-like immunoreactivity in the central nervous system and midgut of the migratory locust. Comparisons with gastrin-cholecystokinin-like and octopamine-like immunoreactivity. Histochemistry 95, 195–203 (1990). https://doi.org/10.1007/BF00266593
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00266593