Abstract
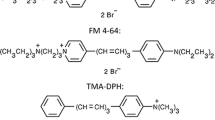
The lateral diffusion of lectin-labelled glycoconjugates was studied in the human colon carcinoma cell line HT29 using fluorescence photobleaching techniques. HT29 cells were grown in either Dulbecco's modified Eagle's medium with glucose (25 mM; DMEM-Glu) or with galactose (25 mM; DMEM-Gal). Cell cultivation in the DMEM-Gal medium was assumed to promote a transformation of the cells to become small-intestinal-like with characteristic microvilli and associated enzymes. The diffusion of glycoconjugates labelled with fluoresceinated Triticum vulgaris agglutinin (Wheat germ agglutinin; WGA), Ricinus communis agglutinin-I (RCA-I), Concanavalia ensiformis agglutinin (ConA), Ulex europaeus agglutinin-I (UEA-I) and Arachis hypogaea agglutinin (PNA) was in all cases rapid, with a diffusion constant (D) ranging between 0.4 and 0.8×10-8 cm2 s-1. As a comparison the diffusion of the fluorescent synthetic lipid analog diI-C14 was characterized by D=0.8 − 1.0 × 10−8 cm2 s-1. The diffusion of lectin-labelled surface components could not be related to the presence of microvilli on HT29 cells grown in DMEM-Gal, which ought to yield an apparently lower diffusion rate. The results indicate either that surface glycoconjugates in HT29 cells are dominated by glycolipid, or that the labelled glycoproteins are more or less free to diffuse in the plane of the membrane.
Similar content being viewed by others
References
Aizenbud BM, Gershon ND (1982) Diffusion of molecules on biological membranes of nonplanar form. A theoretical study. Biophys J 38:287–293
Aizenbud BM, Gershon ND (1984) Diffusion of molecules on microvillous biological membranes. In: Perelson AS, DeLisi C, Wiegel FW (eds) Cell surface dynamics. Concepts and models. Vol 3: Receptors and ligands in intercellular communication. Marcell Dekker, New York Basel, pp 3–21
Axelrod D, Koppel DE, Schlessinger J, Elson E, Webb WW (1976) Mobility measurement by analysis of fluorescence photobleaching recovery. Biophys J 16:1055–1069
Dragsten PR, Blumenthal R, Handler JS (1981) Membrane asymmetry in epithelia: is the tight junction a barrier to diffusion in the plasma membrane? Nature 294: 718–722
Fogh J, Trempe G (1975) New tumor cell lines. In: Fogh J (ed) Human tumor cells “in Vitro”. Plenum Press, New York, pp 115–141
Jacobson K, Derzko Z, Wu E-S, Hou Y, Poste G (1976) Measurement of the lateral mobility of cell surface components in single, living cells by fluorescence recovery after photobleaching. J Supramol Struct 5: 565–56
Lis H, Sharon N (1984) Lectins: properties and applications to the study of complex carbohydrates in solution and on cell surfaces. In: Ginsburg V, Robbins P (eds), Biology of carbohydrates, vol 2. John Wiley, New York, pp 1–86
Magnusson K-E, Wojcieszyn J, Dahlgren C, Stendahl O, Sundqvist T, Jacobson K (1983) Reduced lateral diffusion of wheat germ agglutinin-labelled glycoprotein in the membrane of differentiated HL-60 and U-937 cells assessed with fluorescence recovery after photobleaching (FRAP). Cell Biophys 5: 119–128
Magnusson K-E, Kihlström E, Maluszynska G, Öhman L, Walan A (1986) Biophysical properties of adhesins and other surface antigens. In: Lark D et al. (eds) Protein carbohydrate interactions in biological systems. Academic Press, London, pp 269–274
Peters R (1981) Translational diffusion in the plasma membrane of single cells as studied by fluorescence microphotolysis. Cell Biol Int Rep 5: 733–760
Peters R, Peters J, Tews KH, Bähr W (1974) A microfluorometric study of translational diffusion in erythrocyte membranes. Biochim Biophys Acta 367: 282–294
Peters R, Brunger A, Schulten K (1981) Continuous fluorescence microphotolysis: A sensitive method for study of diffusion processes in single cells. Proc Natl Acad Sci USA 78: 962–966
Pinto M, Appay MD, Simon-Assmann P, Chevalier G, Dracopoli N, Fogh J, Zweibaum A (1982) Enterocytic differentiation of cultured human colon cancer cells by replacement of glucose by galactose in the medium. Biol Cell 44: 193–196
Sjölander A, Magnusson K-E, Latcovic S (1984) The effect of concanavalin A and wheat germ agglutinin on the ultrastructure and permeability of rat intestine. Int Arch Allergy Appl Immunol 75: 230–236
Sjölander A, Magnusson K-E, Latcovic S (1986) Morphological changes of rat small intestine after short-time exposure to concanavalin A or wheat germ agglutinin. Cell Struct Funct 11: 285–294
Szoka F, Jacobson K, Derzko Z, Papahadjopoulos (1980) Studies on the mechanism of liposome-cell interactions in vitro: incorporation of vesicle associated fluorescent probes. Biochim Biophys Acta 600: 1–18
van Meer G, Davoust J, Simons K (1985) Parameters affecting low-pH-mediated fusion of liposomes with the plasma membrane of cells infected with influenza virus. Biochemistry 24: 3593–3602
Zweibaum A, Pinto M, Chevalier G, Dussaulx E, Triadou N, Lacroix B, Haffen K, Brun J-L, Rousset M (1985) Enterocytic differentiation of a subpopulation of the human colon tumor cell line HT-29 selected for growth in sugar-free medium and its inhibition by glucose. J Cell Physiol 122: 21–29
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Magnusson, K.E., Johansson, B. & Sundqvist, T. Rapid lateral diffusion of lectin-labelled glycoconjugates in the human colonic adenocarcinoma cell line HT29. Eur Biophys J 15, 141–145 (1987). https://doi.org/10.1007/BF00263678
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00263678