Summary
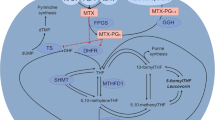
Methotrexate (MTX) accumulates in erythrocytes (ery) during weekly MTX administration, and the ery-MTX concentration reaches a steady state after 4–6 weeks. In order to study MTX accumulation and metabolism to polyglutamate derivatives in different age populations of red blood cells, we took erythrocytes from 12 children with ALL who were receiving maintenance treatment with MTX and 6-MP and separated them according to age on a discontinuous Percoll gradient. When the erythrocytes of these children were separated according to specific gravity a normal distribution was obtained. Age fractionation was confirmed by the exponential decline of the erythrocyte aspartate aminotransferase (ery-ASAT) and by the reticulocyte counts. The ery-MTX declined with increasing red blood cell age in an exponential manner no different from the decline of the ery-ASAT. The youngest population of red blood cells contained 2.3–5.9 (mean 3.8) times more MTX than the oldest population. By linear regression analysis the t1/2 of the ery-MTX was 19–79 days (mean 37 days). The ery-MTX t1/2 seemed to be directly related to the amount of MTX which had been metabolized to MTX-glu3-5. The decline of the ery-MTX was predominantly due to selective disappearance of MTX-glu1+2, whereas MTX-glu3-5 changed to a much lesser extent with advancing red blood cell age. The present investigation showed that steady-state ery-MTX concentration was determined by (1) the amount of MTX added to the circulation by the reticulocytes, (2) the in vivo loss predominantly of MTX with low numbers of glutamyl derivatives from erythrocytes, and (3) the loss of MTX from destroyed red blood cells. The observed in vivo disappearance of MTX from erythrocytes offers a possible explanation of the observation that the ery-MTX steady state was reached after 4–6 weeks of unaltered weekly MTX treatment.
Similar content being viewed by others
References
Balinska M, Galivan J, Coward JK (1981) Efflux of methotrexate and its polyglutamate derivatives from hepatic cells in vitro. Cancer Res 41:2751–2756
Baugh CM, Krumdieck CL, Nair MG (1973) Polygammaglutamyl derivatives of methotrexate. Biochem Biophys Res Commun 52:27–34
Benesh RE, Kwong S, Benesh R, Baugh CM (1985) The binding of folyl and antifolyl polyglutamates to hemoglobin. J Biol Chem 260:14653–14658
Beutler E (1971) Red cell metabolism. A manual of biochemical methods. Grune and Stratton, New York
Costa M da, Iqbal MP (1981) The transport and accumulation of methotrexate in human erythrocytes. Cancer 48:2427–2432
Hendel J (1978) Intracellular metabolites of methotrexate. Chemother Oncol 2 (Suppl): 135–140
Hendel J (1985) Clinical pharmacokinetics of methotrexate in psoriasis therapy. (Thesis) Laegeforeningens, Copenhagen
Hendel J, Nyfors A (1984) Pharmacokinetcs of methotrexate in erythrocytes in psoriasis. Eur J Clin Pharmacol 27:607–610
Jolivet J, Schilsky RL, Bailey BD, Drake DC, Chabner BA (1982) Synthesis, retention and biological activity of methotrexate polyglutamates in cultured human breast cancer cells. J Clin Invest 70:351–360
Kamen BA, Winick NJ (1986) Analysis of methotrexate polyglutamate derivatives in vivo. Methods Enzymol 122:339–345
Kamen BA, Takach PL, Vatev R, Caston JD (1976) A rapid radiochemical ligand binding assay for methotrexate. Anal Biochem 70:54–63
Kamen BA, Nylen PA, Camitta BM, Bertino JR (1981) Methotrexate accumulation and folate depletion in cells as a possible mechanism of chronic toxicity to the drug. Br J Haematol 49:355–360
Kamen BA, Holcenberg JS, Turo K, Whitehead VM (1984) Methotrexate and folate content of erythrocytes in patients receiving oral vs intramuscular therapy with methotrexate. J Pediatr 104: 131–133
Koizumi S, Hurt GA, Fine RL, Griffin JD, Chabner BA (1985) Formation of methotrexate polyglutamates in purified myeloid precursor cells from normal human bone marrow. J Clin Invest 75:1008–1014
Krakower GR, Kamen BA (1983) In situ methotrexate polyglutamate formation in rat issues. J Pharmacol Exp Ther 227:633–638
Krakower GR, Kamen BA (1984) The reticulocytic rat: a model for analysis of methotrexate polyglutamate dynamics in situ. J Pharmacol Exp Ther 231:43–47
Krakower GR, Nylen PA, Kamen BA (1982) Separation and identification of subpicomole amounts of methotrexate polyglutamates in animal and human biopsy material. Anal Biochem 122:412–416
Lindena J, Wittenberg H, Diederichs F, Trautschold I (1986) The decline of catalytic enzyme activty concentration of the in vivo ageing erythrocytes in the man, the dog and the rat. J Clin Chem Clin Biochem 24:49–59
Piomelli S, Corash LM, Davenport DD, Miraglia J, Amorosi EL (1968) In vivo lability of glucose-6-phosphate dehydrogenase in Gde- and GdMediterranean deficiency. J Clin Invest 47:940–948
Salvo G, Caprari P, Samoggia P, Mariani C, Salvati AM (1982) Human erythrocyte separation according to age on a discontinuous Percoll density gradient. Clin Chim Acta 122:293–300
Schalhorn A, Sauer H, Wilmanns W, Stupp-Poutot G (1982) Pharmacokinetics of erythrocyte methotrexate after highdose methotrexate. Cancer Chemother Pharmacol 9:65–69
Schalhorn A, Wilmanns W, Sauer H, Stupp-Poutot G (1985) Methotrexate polyglutamates in human sarcoma tissues and erythrocytes: significance for efficacy of high-dose MTX therapy. Proc Am Assoc Cancer Res 26:235
Scheufler E, Zetler G, Iven H (1981) Pharmacokinetics and organ distribution of methotrexate in the rat. Pharmacology 23:75–81
Schrøder H (1986) Methotrexate pharmacolinetics in agefractionated erythrocytes. Cancer Chemother Pharmacol 18:203–207
Schrøder H (1987) Methotrexate in neutrophils of children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 19:339–342
Schrøder H, Foged E (1986) Methotrexate in erythrocytes of patients with psoriasis. Eur J Clin Pharmacol 30:453–456
Schrøder H, Clausen N, Østergård E, Pressler T (1986) Pharmacokinetics of erythrocyte methotrexate in children with acute lymphoblastic leukemia during maintenance treatment. Cancer Chemother Pharmacol 16:190–193
Schrøder H, Fogh K (1988) Methotrexate and its polyglutamate derivatives in erythrocytes during and after weekly lowdose oral methotrexate therapy of children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 21:145–149
Seaman C, Wyss S, Piomelli S (1980) The decline in energetic metabolism with ageing of the erythrocyte and its relationship to cell death. Am J Hematol 8:31–42
Steele WH, Stuart JFB, Lawrence JR, McNeill CA (1982) The in vivo distribution of methotrexate between plasma and erythrocytes. Cancer Chemother Pharmacol 9:110–113
Winick NJ, Krakower GR, Kamen BA (1986) Metabolism of MTX to polyglutamyl derivatives and the relationship to folate pools in vivo. In: Goldman ID (ed) Proceedings of the Second Workshop on Folyl and Antifolyl Polyglutamates. Praeger Scientific, London, New York, p 297
Author information
Authors and Affiliations
Additional information
The present work was supported by grant no. 12-6387 from the Danish Medical Research Council and by grants from the Institute of Experimental and Clinical Research, Universty of Aarhus Denmark
Rights and permissions
About this article
Cite this article
Schrøder, H., Fogh, K. & Herlin, T. In vivo decline of methotrexate and methotrexate polyglutamates in age-fractionated erythrocytes. Cancer Chemother. Pharmacol. 21, 150–155 (1988). https://doi.org/10.1007/BF00257363
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00257363