Summary
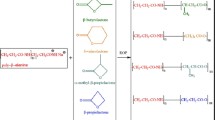
The α-phenyl-α-alkyl-β-propiolactones (alkyl groups =-CH2CH3; -CH2CH2CH3; -CH2CH2CH2CH3) were synthesized and polymerized by anionic ring-opening polymerization by tetra-ethylammonium benzoate (non-chiralic initiator). It was found that the molecular weight of polylactones increased as the size of the alkyl substituent increased. Poly(α-propyl-α-phenyl-β-propiolactone) showed the highest melting temperature. Lactones polymerized using tetraethylammonium dibenzoiltartrate (chiralic initiator) gave polyesters with higher melting temperature than those obtained with a non-chiralic initiator under the same conditions.
Similar content being viewed by others
References
ALLEGREZZA A.E, LENZ R.W, CORNIBERT J, MARCHESSAULT R.H, J. Polym. Sci, Polym. Chem. Ed. 16, 2617 (1978)
BIGDELI E, LENZ R.W, Macromolecules 11, 493 (1978)
BORRI C, BRUCKNER S, Eur. Polymer J. 7, 1975 (1971)
CARRIERE F.J, EISENBACH C.D, Makromol. Chem. 182, 325 (1981)
CORNIBERT J, MARCHESSAULT R.H, ALLEGREZZA A.E, LENZ R.W, Macromolecules 6, 676 (1973)
D'HONT CH, LENZ R.W, J. Polym. Sci., Polym. Chem. Ed. 16, 261
EISENBACH C.D, LENZ R.W, Makromol. Chem. 177, 2539 (1976)
HALL JR, A.K., Makromolecules 2, 488 (1969)
LENZ R.W, Glas. Hem. Drus. Beograd 39, 395 (1974)
MARCHESSAULT R.H, PIERRE J. ST, DUVAL M, PEREZ S, Macromolecules 11, 1281 (1978)
ROBERTS J.D, MC MAHOM W. and MC MAHOM R.E., J. Amer. Chem. Soc. 73, 763 (1951)
ROBERTS R.C, J. Polymer Sci. B 8, 381 (1970)
TESTA E, FONTANELLA L, FAVA F, Justus Liebigs Ann. Chem. 619, 47 (1958a), id. 614, 158 (1958b)
TESTA E, FONTANELLA L, FAVA F, Farmaco Ed. Sci. 13, 152 (1958c)
TESTA E, FONTANELLA L, Justus Liebigs Ann. Chem. 614, 167 (1958d)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Canessa, G., Pooley, S.A., Rodriguez-Baeza, M. et al. Racemic poly(α-alkyl-α-phenyl-βpropiolactone) substituent dependence. Polymer Bulletin 6, 515–519 (1982). https://doi.org/10.1007/BF00256372
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00256372