Summary
For the first time, the polymolybdates of organic bases have been synthesized (BH)4Mo8O26 (B = 2-aminopyridine, 3-aminopyridine and 4-aminopyridine). These compounds have been characterized using X-ray and infrared spectroscopy. The energies of thermal decomposition have been determined by differential calorimetry.
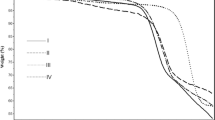
On the other hand, the molecular weights of these new compounds in DMSO solution have been measured by vapour pressure osmometry and viscometry, obtaining results in agreement with the structures, as well as with the activation energies corresponding to the thermal decomposition, calculated from the TGA curves.
Similar content being viewed by others
References
GATEHOUSE,B.M. and LEVERETT,P.: Chem.Comm., 740(1970)
GATEHOUSE,B.M. and LEVERETT,P.: J. Chem. Soc.(A), 2107 (1971)
KATIME,I. and AGUILAR,F.: An.Química (in press)
PERRIN,D.D.:Dissociation Constants of Organic Bases in Aqueous Solution, London, Butterworths 1965
ROMAN,P.: Ph.D. Thesis. Bilbao University 1976
TELLO,M.J., BOCANEGRA,E.H., GILI,P., LORENTE,L. and ROMAN,P.: Thermochim. Acta,12, 65 (1975)
TELLO,M.J., LORENTE,L., ROMAN,P., GILI,P. and SANTIAGO,C.: Thermochim. Acta, 21, 135 (1977)
SATAVA,V.: Thermochim. Acta, 2, 423 (1971)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gili, P., Roman, P. & Katime, I.A. Inorganic polymers. Polymer Bulletin 1, 509–515 (1979). https://doi.org/10.1007/BF00255716
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00255716