Summary
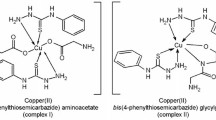
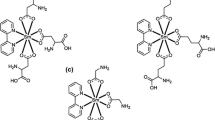
The title monomer has been prepared for the first time. It has been polymerized by a free radical mechanism in different solvents. Only oligomers were obtained so far. The dependence of the rate of polymerization on monomer and initiator concentrations has been measured and was found to follow the usual free radical scheme. The overall activation energy of polymerization has been measured in two solvents and was found to be 21.2 and 15.03 K.Cal/mol in tetrahydrofuran (THF) and N,N-dimethyl formamide (DMF) respectively. The prepared monomer has excellent chelating ability with different metal cations. The stability constant of the monomer with copper (II) ions has been measured in acetone as well as in dioxane. The complexes have no tendency for free radical polymerization.
Similar content being viewed by others
References
COWIE J.M.G., and WADI N.M., Polymer, 26, 1566 (1985).
FOYE WILLIAM O., and CHAO CHING CHIU, J.Pharm.Sci., 73 (9), 1284 (1984).
GUPTA P., CHAUDHARY R.S., and PRAKASH B., J.Electrochem. Soc. India, 32 (4), 406 (1083).
KANEKO M., and TSUCHIDA E., J. Polym. Sci Macromolecular Reviews, 16, 397 (1981).
SAHA B.G., and BANERJI S.K., Indian J. Technol. 21(7) 277, (1983).
TSUCHIDA E., and NISHIDE H., Adv. Polym. Sci., 24, 1 (1977).
VALIGURA D., and ONDREJOVIC G., Proc. Conf. Coord., Chem., 10th, 445 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Elsabee, M.Z., Sabaa, M.W., Naguib, H.F. et al. Polymerization and chelating behavior of N-acryloyl, N-phenylthiourea. Polymer Bulletin 22, 143–149 (1989). https://doi.org/10.1007/BF00255204
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00255204