Summary
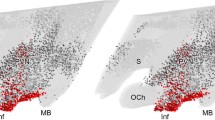
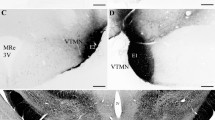
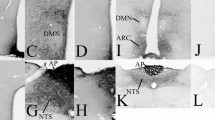
The in vivo radioautographic method has been applied to elucidate the mechanism of direct peptide hormone “feedback” action in the CNS. Using this method we have identified the circumventricular organs of the brain as general endocrine target tissues for a variety of blood-borne polypeptide hormones, including insulin. In the arcuatemedian eminence region of the hypothalamus blood-borne insulin directly interacts with receptive nerve terminals, suggesting that insulin acts to influence the electrical activity of select hypothalamic nerve circuits at the level of synaptic transmission. Recent results obtained from preliminary surgical and chemical lesion studies of brain indicate that insulin-receptive nerve terminals in the arcuate-median eminence region arise from neurons intrinsic to the medial basal hypothalamus. This has lead us to propose the concept of the hypothalamic tuberoinfundibular insulin-receptive neuron and its axon collaterals as a pathway for the centripetal flow of insulin “signals” in the form of electrical impulses. We envisige that the neuroanatomic pathway, provided by the hypothalamic tuberoinfundibular neuron, functions to link changes in body metabolic activity, as reflected in changing levels of circulating insulin, to the neuronal process of elaborating specific central metabolic-regulatory programs. This pathway could be of key importance in understanding and combating metabolic disease.
Article PDF
Similar content being viewed by others
References
Anand BK, Chhina GS, Sharma KN, Dua S, Singh B (1964) Activity of single neurons in the hypothalamic feeding centers: effect of glucose. Am J Physiol 207: 1146–1154
Arieff AI, Doerner T, Zelig H, Massry SG (1974) Mechanisms of seizure and coma in hypoglycemia. Evidence for a direct effect of insulin on electrolyte transport in brain. J Clin Invest 54: 654–663
Bergeron JJM, Posner BI (1979) In vivo studies on the initial localization and fate of polypeptide hormone receptors by the technique of quantitative radioautography. J Histochem Cytochem 27: 1512–1515
Bernardis LL, Frohman LA (1971) Effects of hypothalamic lesions at different loci on development of hyperinsulinemia and obesity in the weanling rat. J Comp Neurol 141: 107–115
Berthoud H-R, Jeanrenaud B (1979) Acute hyperinsulinemia and its reversal by vagotomy after lesions of the ventromedial hypothalamus in anesthetized rats. Endocrinology 105: 146–151
Broadwell RD, Brightman MW (1976) Entry of peroxidase into neurons of the central and peripheral nervous systems from extracerebral and cerebral blood. J Comp Neurol 166: 257–284
Brobeck JR, Tepperman J, Long CHN (1943) Experimental hypothalamic hyperphagia in the albino rat. Yale J Biol Med 15: 831–853
Cain DP (1975) Effects of insulin injection on responses of olfactory bulb and amygdaloid single units to odors. Brain Res 99: 69–81
Chen M, Woods SC, Porte D Jr (1975) Effect of cerebral intraventricular insulin on pancreatic insulin secretion in the dog. Diabetes 24: 910–914
Debons AF, Krimsky I, From A, Clouttier RJ (1969) Rapid effects of insulin on the hypothalamic satiety center. Am J Physiol 217: 1114–1119
Debons AF, Krimsky I, From A (1970) A direct action of insulin on the hypothalamic satiety center. Am J Physiol 219: 938–943
De Jong A, Strubbe JH, Steffens AB (1977) Hypothalamic influence on insulin and glucagon release in the rat. Am J Physiol 233: E280–288
Freychet P, Roth J, Neville DM Jr (1971) Insulin receptors in the liver: Specific binding of 125I-insulin to the plasmalemma and its relation to insulin bioactivity. Proc Natl Acad Sci USA 68: 1833–1837
Frohman LA, Bernardis LL, Kant KJ (1968) Hypothalamic stimulation of growth hormone release. Science 162: 580–582
Frohman LA, Bernardis LL (1971) Effect of hypothalamic stimulation on plasma glucose, insulin and glucagon levels. Am J Physiol 221: 1596–1603
Frohman LA, Muller EE, Cocchi D (1973) CNS-mediated inhibition of insulin secretion due to 2-deoxyglucose. Horm Metab Res 5: 21–26
Fuxe K, Andersson K, Hokfelt T, Agnati LF, Ogren S-O, Eneroth P, Gostafson J-A, Skett P (1978) Prolactin-monoamine interactions in rat brain and their importance in regulation of LH and prolactin secretion. In: Robyn R, Harter J (eds) Progress in prolactin physiology and pathology. Elsevier North Holland Medical Press, Amsterdam, p 94–109
Halasz B, Pupp L (1965) Hormone secretion of anterior pituitary gland after physical interruption of all nervous pathways to the hypophysiotropic area. Endocrinology 77: 533–562
Harper AE (1976) Protein and amino acids in regulation of food intake. In: Novins D, Wyrwicka W, Bray G (eds) Hunger: Basic mechanisms and clinical implications. Raven Press, New York, p 103–113
Hatfield JS, Millard WJ, Smith CJV (1974) Shortterm influence of intraventromedial hypothalamic administration of insulin in normal and diabetic rats. Pharmacol Biochem Behav 2: 223–226
Havrankova J, Brownstein M, Roth J (1978) Insulin receptors are widely distributed in the CNS of the rat. Nature 272: 827–829
Hoebel GB (1971) Feeding: neural control of intake. Annu Rev Physiol 33: 533–568
Junger E (1978) Autoradiographic demonstration of insulin binding sites on cell surfaces. Cytobiologie 18: 250–258
Jonsson G, Fuxe K, Hokfelt T (1972) On the catecholaminergic innervation of the hypothalamus with special reference to the median eminence. Brain Res 40: 271–281
Kahn CR (1976) Membrane receptors for hormones and neurotransmitters. J Cell Biol 70: 261–286
Kennedy GC (1953) The role of depot fat in the hypothalamic control of food intake in the rat. Proc R Soc B 140: 549–553
Kennedy GC (1966) Food intake, energy balance and growth. Br Med Bull 22: 216–220
Kennedy GC (1969) Interactions between feeding, behavior and hormones during growth. Ann NY Acad Sci 157: 1049–1061
Kerr FWL, Preshaw RM (1969) Secretomotor function of the dorsal motor nucleus of the vagus. J Physiol (Lond) 205: 405–418
Kimura H, Kuriyama K (1975) Distribution of gamma ammobutyric acid (GABA) in the rat hypothalamus: functional correlates of GABA with activity of appetite controlling mechanisms. J Neurochem 24: 903–907
Lund-Andersen H (1979) Transport of glucose from blood to brain. Physiol Rev 59: 305–358
Marrazzi MA (1976) Hypothalamic glucoreceptor response — Biphasic nature of unit potential changes. In: Novin D, Wyrwicka W, Bray G (eds) Hunger: Basic mechanisms and clinical implications. Raven Press, New York, p 171–178
Mayer J (1955) Regulation of energy intake and body weight: Glucostatic and lipostatic hypothesis. Ann N Y Acad Sci 63: 15–42
McCaleb ML, Myers RD, Singer G, Willis G (1979) Hypothalamic norepinephrine in the rat during feeding and pushpull perfusion with glucose, 2-DG or insulin. Am J Physiol 236: R 312–321
Meeker R, Myers RD (1979) In vivo (14C) amino acid profiles in discrete hypothalamic regions during push-pull perfusion in the unrestrained rat. Neurosciences 4: 495–506
Olney JW, Schwainker B, Rhee V (1976) Chemical lesioning of the hypothalamus as a means of studying neuroendocrine function. In: Sacher EJ (ed) Hormones, behavior and psychopathology. Raven Press, New York, p 153–158
Oomura Y, Ooyama H, Yamamoto T, Ono T, Kobayashi N (1969) Behaviour of hypothalamic units during electrophoretic application of drugs. Ann NY Acad Sci 157: 642–665
Oomura Y (1976) Significance of glucose, insulin and free fatty acids on the hypothalamic feeding and satiety neurons. In: Novin D, Wyrwicka W, Bray G (eds) Hunger: Basic mechanisms and clinical implications. Raven Press, New York, p 145–157
Pacold ST, Blackard WG (1979) Central nervous system insulin receptors in normal and diabetic rats. Endocrinology 105: 1452–1457
Palkovits M, Léranth Cs, Zaborszky L, Brownstein MJ (1977) Electron microscope evidence of direct neuronal connections from the lower brain stem to the median eminence. Brain Res 136: 339–344
Palkovits M (1979) Effect of surgical deafferentation on the transmitter and hormonal content of the hypothalamus. Neuroendocrinology 29: 140–148
Panksepp J (1972) Hypothalamic radioactivity after intragastric glucose-14C in rats. Am J Physiol 223: 396–401
Panksepp J (1974) Hypothalamic regulation of energy balance and feeding behavior. Fed Proc 33: 1150–1165
Panksepp J (1975) Medial and lateral hypothalamic oxygen consumption as a function of age, starvation and glucose administration in rats. Brain Res 94: 133–140
Pecile A, Muller EE, Felici M, Netti C (1971) Nervous system participation in growth hormone release from the anterior pituitary gland. In: Pecile A, Muller EE (eds) Growth and growth hormone. Excerpta Medica, Amsterdam, p 261–282
Phillips MI (1978) Angiotensin in the brain. Neuroendocrinology 25: 354–377
Powley TL (1977) The VMH syndrome, satiety and a cephalic phase hypothesis. Psychol. Rev 84: 89–126
Renaud LP, Martin JB (1975) Electrophysiologic studies of connections of the hypothalamic ventromedial nucleus of the rat: evidence for a role in neuroendocrine regulation. Brain Res 93: 145–151
Renaud LP, Martin JB (1976) Tuberoinfundibular neurons in the basomedial hypothalamus of the rat: electrophysiologic evidence for axon collaterals to hypothalamic and extrahypothalamic areas. Brain Res 105: 59–72
Ricardo JA, Koh ET (1978) Anatomical evidence of direct projections from the nucleus of the solitary tract to the hypothalamus, amygdala and other forebrain structures in the rat. Brain Res 153: 1–26
Rohner F, Dufour A-C, Karakash C, Le Marchand Y, Ruf KB, Jeanrenaud B (1977) Immediate effect of lesion of the ventromedial hypothalamic area upon glucose-induced insulin secretion in anesthetized rats. Diabetologia 13: 239–242
Shimazu T, Fukuda A, Ban T (1966) Reciprocal influence of the ventromedial and lateral hypothalamic nuclei on blood glucose levels and liver glycogen content. Nature 210: 1178–1179
Shimazu T, Ogasawara S (1975) Effects of hypothalamic stimulation on gluconeogenesis and glycolysis in rat liver. Am J Physiol 226: 1787–1793
Storlien LH, Bellingham WP, Martin GM (1975) Localization of CNS glucoregulatory insulin receptors within the VMH. Brain Res 96: 156–160
Szabo O, Szabo AJ (1972) Evidence for an insulin-sensitive receptor in the CNS. Am J Physiol 223: 1349–1353
Szabo O, Szabo AJ (1975) Studies on the nature and mode of action of the insulin-sensitive glucoreceptor in the CNS. Diabetes 24: 328–336
Tappaz ML, Brownstein MJ (1977) Origin of glutamate decarboxylase (GAD) containing cells in discrete hypothalamic nuclei. Brain Res 132: 95–106
Tappaz M, Aguera M, Belin MF, Pujol JF (1980) Autoradiography of gamma aminobutyric acid in the rat median eminence. Brain Res 186: 379–391
van Houten M, Posner BI (1979) Insulin binds to brain blood vessels in vivo. Nature 282: 623–625
van Houten M, Posner BI, Kopriwa BM, Brawer JR (1979) Insulin binding sites in the rat brain: in vivo localization to the circumventricular organs by quantitative radioautography. Endocrinololgy 105: 666–673
van Houten M, Posner BI, Kopriwa BM, Brawer JR (1980) Insulin binding sites localized to nerve terminals in rat median eminence and arcuate nucleus. Science 207: 1081–1083
van Houten M, Posner BI, Walsh RJ (1980) Radioautographic identification of lactogen binding sites in the rat median eminence using 125I-human growth hormone: evidence for a prolactin “short-loop” feedback site. Exp Brain Res 38: 455–461
van Houten M, Schiffrin EL, Mann JFE, Posner BI, Boucher R (1980) Radioautographic localization of specific binding sites for blood-borne angiotensin II in the rat brain. Brain Res 186: 480–485
van Houten M, Posner BI (1980) Brain insulin receptors: radioautographic evidence for rapid internalization within neurons of the rat area postrema. Endocrine Society Abstracts A 176.8
van Houten M, Goltzman D, Posner BI (1980) Brain calcitonin receptors: Localization in normals and deficiency in hereditary diabetes insipidus. Clin Res 28: 676 A
Weindl A (1973) Neuroendocrine aspects of the circumventricular organs. In: Ganong WF, Martini L (eds) Frontiers in neuroendocrinology. Oxford Press, New York, p 1–32
Wirtschafter D, Davis JD (1977) Body weight: reduction by long-term glycerol treatment. Science 198: 1271–1274
Woods SC, Porte D Jr (1974) Neural control of the endocrine pancreas. Physiol Rev 54: 596–619
Woods SC, Decke E, Vasselli JR (1974) Metabolic hormones and regulation of body weight. Psychol Rev 81: 126–143
Woods SC, Porte D Jr (1975) Effect of intracisternal insulin on plasma glucose and insulin in the dog. Diabetes 24: 905–909
Woods SC, Porte D Jr (1978) The CNS, pancreatic hormones, feeding and obesity. In: Advances in metabolic disorders, vol 9. Academic Press, New York, p 283–311
Woods SC, Lotter EC, McKay LD, Porte D Jr (1979) Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 282: 503–505
Wurtman RJ (1976) Control of neurotransmitter synthesis by precursor availability and food consumption. In: Naftolin F, Ryan KJ, Davies J (eds) Subcellular mechanisms in reproductive neuroendocrinology. Elsevier Publishing Comp, Amsterdam p 146–166
Yagi K, Sawaki Y (1974) Recurrent inhibition and facilitation: Demonstration of the tuberoinfundibular system and effects of strychnine and picrotoxin. Brain Res 84: 155–159
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van Houten, M., Posner, B.I. Cellular basis of direct insulin action in the central nervous system. Diabetologia 20 (Suppl 1), 255–267 (1981). https://doi.org/10.1007/BF00254491
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00254491