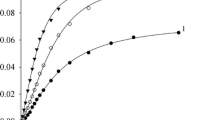
Abstract
The reaction of 1,1-diphenylethylene with AlCl3 in CH2Cl2 at low temperature in the presence of small amounts of water (present as residual impurity or deliberately added) occurs in two successive stages: a very rapid reaction cocatalysed by water and a slower one resulting from a direct (bimolecular) initiation. The stoichiometry of the reaction involving water has been determined: 3 H2O and 6 AlCl3, are necessary to form 4 carbocations.
In the same conditions, hydrogen chloride has no influence either on the extent of the first stage or on the yield and the rate of the second stage.
Similar content being viewed by others
References
G. SAUVET, J.P. VAIRON and P. SIGWALT, J. Polymer Sci., Symposium n∘ 52, 173 (1975)
M. MASURE, G. SAUVET and P. SIGWALT, J. Polymer Sci., 16, 3065 (1978)
G. SAUVET, J.P. VAIRON and P. SIGWALT, J. Polym. Sci., 16, 3047 (1978)
Y.Y. FIALKOV, A.P. SHUTKO and P. Ya. MUUR, Khim. Tekhnol., 58 (1973)
A. STORR, K. JONES and A.W. LAUBENGAYER, J. Amer. Chem. Soc., 90, 3173 (1968)
A. FELTZ, Z. Anorg. Allgem. Chem., 332, 35 (1964)
H.C.BROWN and H. PEARSALL, J. Amer. Chem. Soc., 73, 4681 (1957)
D.O. JORDAN and A.R. MATHIESON, “Cationic Polymerization and Related complexes”, P.H. Plesch Ed., 91 (1953)
J.P. KENNEDY and R.G. SQUIRES, J. Macromol. Sci. Chem. A1, 995 (1967)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Masure, M., Sauvet, G. & Sigwalt, P. Effects of water and hydrogen chloride on the reaction of 1,1-diphenylethylene with AlCl3 . Polymer Bulletin 2, 699–705 (1980). https://doi.org/10.1007/BF00254455
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00254455