Abstract
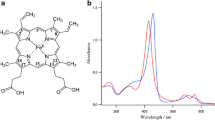
The dispersion of the depolarization ratio of oxidation- and spin-marker lines of sperm whale myoglobin derivatives (oxyMb, deoxyMb, ferric Mb-CN) and of ferric Hb-CN have been measured for different pH-values in the acid and alkaline region. No pH-dependence in the region above pH=6.5 has been found. Below pH=6.5, however, a significant pH-dependence of the oxyMb-oxidation marker line at 1,375 cm-1 exists. Additionally, a weak pH-dependence of the corresponding 1,355 cm-1 line of the deoxymyoglobin spectrum is observed. This effect can be explained assuming a titration of distal histidine, inducing a rupture of the ligand-imidazole H-bond in the case of oxymyoglobin. The pH-independent depolarization ratio disperson above pH=6.5 in all systems investigated is explained by the lack of the haemoglobin saltbridge between His(HC3)β and Asp(FG5)β, which is essential for the cooperativity in the haemoglobin system.
Similar content being viewed by others
Abbreviations
- DPR:
-
depolarization ratio
- EP:
-
excitation profile
- oxyMb:
-
oxymyoglobin
- deoxyMb:
-
deoxymyoglobin
- metMb-CN:
-
ferric myoglobincyanide
- metHb-CN:
-
ferric haemoglobincyanide
References
Anusiem AC, Beetlestone JG, Irvine DH (1968) Reactivity differences between haemoglobins. Part VIII. The thermodynamics of the reaction of human methaemoglobins A and C with fluoride, thiocyanate, and cyanide ions. An interpretation of enthalpy changes in terms of hydration. J Chem Soc (A): 960–969
Blumenthal DC, Kassner RJ (1980) Cyanide binding to the cytochrome c ferric heme octapeptide. A model for anion binding to the active site of high spin ferric heme proteins. J Biol Chem 255: 5859–5863
Christahl M, Gersonde K (1982) Structure-related changes of the electron spin resonance spectra of the monomeric nitrosyl haemoglobin IV from Chironomus thummi thummi. Biophys Struct Mech 8: 271–288
Deatherage JF, Loe RS, Anderson CM, Moffat K (1976) Structure of cyanide methemoglobin. J Mol Biol 104: 687–706
Deb SK, Bausol ML, Roy AP (1984) Calculation of error in depolarization ratio measurements due to finite colection angle in laser raman spectroscopy. Appl Spectrosc 98: 500–504
Gibson QH, Parkhurst LJ, Geraci G (1969) The reaction of methemoglobin with some ligands. J Biol Chem 244: 4668–4676
Heidner EJ, Ladner RC, Perutz MF (1976) Structure of horse carbonmonoxyhaemoglobin. J Mol Biol 104: 707–722
Kendrew JC, Parrish RG (1956) The crystal structure of myoglobin III. Sperm-whale myoglobin. Proc R Soc (London) A 238: 305–324
Kilmartin JV, Fogg JH, Perutz MF (1980) Role of C-terminal histidine in the alkaline Bohr effect of human hemoglobin. Biochemistry 19: 3189–3193
La Mar GM, Budd DL, Sick H, Gersonde K (1978) Acid Bohr effects in myoglobin characterized by proton NMR hyperfine shifts and oxygen binding studies. Biochim Biophys Acta 537: 270–283
Ohms JP, Hagenmaier H, Hayes MB, Cohen JS (1979) Near-heme histidine residues of deoxy- and oxymyoglobins. Biochemistry 18: 1599–1602
Parak F, Kalvius GM (1982) Anwendung des Mößbauereffektes auf Probleme der Biophysik. In: Hoffe F, Lohmann W (Hrsg) Biophysik, 2nd edn. Springer, Berlin Heidelberg New York, pp 159–183
Perutz MF (1970a) Stereochemistry of cooperative effects in haemoglobin. Nature 228: 726–734
Perutz MF (1970b) The Bohr effect and combination with organic phosphates. Nature 228: 734–739
Russu IM, Ho NT, Ho C (1980) Role of the β146 histidyl residue in the alkaline Bohr effect of hemoglobin. Biochemistry 19: 1043–1052
Russu IM, Ho NT, Ho C (1982) A proton nuclear magnetic resonance investigation of histidyl residues in human normal adult hemoglobin. Biochemistry 21: 5031–5043
Schweitzer R, Dreybrodt W, Mayer A, el Naggar S (1982) Influence of the solvent environment on the polarization properties of resonance raman scattering in haemoglobin. J Raman Spectrosc 13: 139–147
Schweitzer-Stenner R, Dreybrodt W, el Naggar S (1984a) Investigation of pH-induced symmetry distortions of the prosthetic group in deoxyhaemoglobin by resonance raman scattering. Biophys Struct Mech 10: 241–256
Schweitzer-Stenner R, Dreybrodt W, Wedekind D, el Naggar S (1984b) Investigation of pH-induced symmetry distortions of the prosthetic group in oxyhaemoglobin by resonance raman scattering. Eur Biophys J 11: 61–76
Shaanan B (1982) The iron-oxygen bond in human oxyhaemoglobin. Nature 296: 683–684
Simon SR, Arndt DJ, Königsberg WH (1971) Structure and functional properties of chemically modified horse hemoglobin. J Mol Biol 58: 69–77
Spiro TG, Strekas TC (1974) Resonance raman spectra of hemeproteins. Effects of oxidation and spin state. J Am Chem Soc 96: 338–345
Takano T (1977) Structure of myoglobin refined at 2.0 Å resolution. J Mol Biol 110: 537–568
Wedekind D, el Naggar S, Schweitzer-Stenner R, Dreybrodt W (1984) PH-induced changes of tertiary structure of oxy-HbA detected by resonance raman spectroscopy. Proceedings of the 9th International Symposium on Raman Spectroscopy and Biological Sciences, Osaka, Japan, 24. 8.–25. 8. 1984
Wilbur DJ, Allerhand A (1977) Titration behaviour and tautomeric states of individual histidine residues of myoglobin. J Biol Chem 252: 4968–4975
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
el Naggar, S., Dreybrodt, W. & Schweitzer-Stenner, R. Haem-apoprotein interactions detected by resonance Raman scattering in Mb- and Hb-derivates lacking the saltbridge His146β-Asp94β. Eur Biophys J 12, 43–49 (1985). https://doi.org/10.1007/BF00254094
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00254094