Abstract
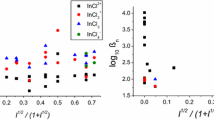
The interaction between lead(II) and methanesulfonate ions was studied by conventional d.c. polarography at 2.0 M ionic strength and pH 4.0. Two stepwise lead complexes are formed in solution: [Pb(CH3SO3)]+, ß1 = 1.76 ± 0.03 M−1 and [Pb(CH3SO3)2], β2 = 0.25 ± 0.05 M−2 which are reversibly reduced at the DME. At high ligand concentrations, above 1.0 M, a third lead complex, [Pb(CH3SO3)3] 433= 0.30±0.07M −3, is formed.
Similar content being viewed by others
References
G. Leuchs, D.R. Patent, 38193 (1886).
H. Benninghoff, Galvanotechnik 57 (1966) 168.
J. A. von Fraunhofer, ‘Basic Metal Finishing’, Elek Science, London (1976).
A. T. Khun (ed), ‘The Electrochemistry of Lead’, Academic Press, London (1979).
C. Rosenstein, Metal Finish. 88 (1990) 17.
C. A. Wamser, J. Am. Chem. Soc. 70 (1948) 1209.
R. E. Mesmer, K. M. Palen and C. F. Baes, Inorg. Chem. 12 (1973) 89.
J. P. Candlin and R. G. Wilkins, J. Chem. Soc. (1960) 4236.
R. B. Fisher and P. Hafey, Proc. Indiana Acad. Sci. 72 (1972) 123.
J. M. Notley, J. Appl. Chem. Biotechnol. 23 (1973) 717.
W. A. Proell, C. E. Adams and B. H. Shoemaker, Ind. Eng. Chem. 40 (1948) 1129.
Ibid. US Patent 2525942 (1950).
Daiwa Chemicals Co., Ltd. and Ishihara Chemical Co., Ltd., Fr. Patent 2534279 (1984); Eur. Patent 192273 (1986).
K. Owata and N. Dohi, Jpn. Kokai Tokkio Koho 59182986 (1984).
Mitsubishi Electr. Corp., ibid. 59 211562 (1984).
Daiwa Fine Chemicals Co., Ltd., U.S. Patent 4555314 (1985).
V. C. Opskar and G. S. Bokisa, Eur. Patent EP 207732 (1985).
U. Opskar, V. Canaris and W. J. Wiliam, U.S. Patent 4582576 (1986).
Daiwa Kasei Kenkyusho K. K. and Ishihaara Yakuhin Co., Ltd., Jpn. Kokai Tokkio Koho 6148 589 (1986).
S. Matsumoto, Y. Nakano, D. Tonai and M. Matsuda, ibid. 61194196 (1986).
V. M. Sajja, R. Mathew and J. Belane, US Patent 4589962 (1986).
W. L. Hsueh and C. C. Wan, Bull. Electrochem. 6 (1990) 790.
L. Meites, ‘Polarographic Techniques’, 2nd. edn., Interscience, New York (1965).
D. D. DeFord and D. N. Hume, J. Am. Chem. Soc. 73 (1953) 5321.
D. N. Hume, D. D. DeFord and G. C. B. Cave, ibid. 73 (1953) 5323.
E. A. Burns and D. N. Hume, ibid. 78 (1956) 3958.
J. C. Ringen and R. E. Kirby, J. Inorg. Nucl. Chem. 36 (1974) 199.
A. Kumar and J. N. Gaur, J. Electroanal. Chem. 59 (1974) 317.
K. B. Yatsmirskii and P. Vasil'ev, ‘Instability Constants of Complex Compounds’, Enterprises, New York (1960).
M. T. Beck, ‘Chemistry of Complex Compounds’, Van Nostrand, London (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Capelato, M.D., Nóbrega, J.A. & Neves, E.F.A. Complexing power of alkanesulfonate ions: the lead-methanesulfonate system. J Appl Electrochem 25, 408–411 (1995). https://doi.org/10.1007/BF00249661
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00249661