Abstract
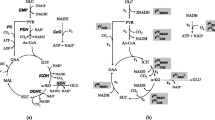
Corynebacterium callunae (NCIB 10338) grows faster on glutamate than ammonia when used as sole nitrogen sources. The levels of glutamine synthetase (GS; EC 6.3.1.2) and glutamate synthase (GOGAT; EC 1.4.1.13) of C. callunae were found to be influenced by the nitrogen source. Accordingly, the levels of GS and GOGAT activities were decreased markedly under conditions of ammonia excess and increased under low nitrogen conditions. In contrast, glutamate dehydrogenase (GDH; EC 1.4.1.4) activities were not significantly affected by the type or the concentration of the nitrogen source supplied. The carbon source in the growth medium could also affect GDH, GS and GOGAT levels. Of the carbon sources tested in the presence of 2 mM or 10 mM ammonium chloride as the nitrogen source pyruvate, acetate, fumarate and malate caused a decrease in the levels of all three enzymes as compared with glucose. GDH, GS and GOGAT levels were slightly influenced by aeration. Also, the enzyme levels varied with the growth phase. Methionine sulfoximine, an analogue of glutamine, markedly inhibited both the growth of C. callunae cells and the transferase activity of GS. The apparent K m values of GDH for ammonia and glutamate were 17.2 mM and 69.1 mM, respectively. In the NADPH-dependent reaction of GOGAT, the apparent K m values were 0.1 mM for α-ketoglutarate and 0.22 mM for glutamine.
Similar content being viewed by others
Abbreviations
- GDH:
-
glutamate dehydrogenase
- GS:
-
glutamine synthetase
- GOGAT:
-
glutamate synthase
References
Adachi K, Suzuki I (1977) Purification and properties of glutamate synthase from Thiobacillus thioparus. J Bacteriol 129: 1173–1182
Bernlohr RW, Schreier HJ, Donohue TJ (1984) Enzymes of glutamate and glutamine biosynthesis in Bacillus licheniformis. Curr Top Cell Regul 24: 145–152
Bohannon DE, Rosenkrantz MS, Sonenshein AL (1985) Regulation of Bacillus subtilis glutamate synthase genes by the nitrogen source. J Bacteriol 163: 957–964
Brown CM (1980) Ammonia assimilation and utilization in bacteria and fungi. In: Payne JW (ed) Microorganisms and nitrogen sources. Wiley & Sons. Chichester New York Brisbane, pp 511–535
Caballero FJ, Igeno I, Cardenas J, Castillo F (1989) Regulation of reduced nitrogen assimilation in Rhodobacter capsulatus E1F1. Arch Microbiol 152: 508–511
Ertan H (1992) Some properties of glutamate dehydrogenase, glutamine synthetase and glutamate synthase from Corynebacterium callunae. Arch Microbiol 158: 35–41
Fisher SH, Rosenkrantz MS, Sonenshein AL (1984) Glutamine synthetase gene of Bacillus subtilis. Gene 32: 427–438
Harder W, Dijkhuizen L (1983) Physiological responses to nutrient limitation. In: Ornston LN, Balows A, Baumann P (eds) Annual review of microbiology, vol 37. Annual Review Inc., California, pp 1–25
Hemmila IA, Mantsala PI (1978) Purification and properties of glutamate synthase and glutamate dehydrogenase from Bacillus megaterium. Biochem J 173: 45–52
Hirose Y, Okada H (1979) Microbial production of amino acids. In: Pepler HJ, Perlman D (eds) Microbial technology, 2nd edn, vol 1. Academic Press, New York San Francisco London, pp 211–241
Howitt SM, Gresshoff PM (1985) Ammonia regulation of glutamine synthetase in Rhizobium sp. ANU289. J Gen Microbiol 131: 1433–1440
Jenkinson HF, Buttery PJ, Lewis D (1979) Assimilation of ammonia by Bacteroides amylophilus in chemostat cultures, J Gen Microbiol 113: 305–313
Kanamori K, Weiss RL, Roberts JD (1987) Ammonia assimilation in Bacillus polymyxa: 15N NMR and enzymatic studies. J Biol Chem 262: 11038–11045
Kim C-H, Hollocher TC (1982) 13N Isotope studies on the pathway of ammonia assimilation in Bacillus megaterium and Escherischia coli. J Bacteriol 151: 358–366
Kinoshita S (1985) Glutamic acid bacteria. In: Demain AL, Solomon NA (eds) Biology of industrial microorganisms. Benjamin/Cummings Publishing, London Amsterdam Don Mills, pp 115–143
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 165–275
Madigan M, Cox SS (1982) Nitrogen metabolism in Rhodopseuomonas globiformis. Arch Microbiol 133: 6–10
Magasanik B, Prival MJ, Brenchley JE (1973) Glutamine synthetase, regulator of the synthesis of glutamate-forming enzymes. In: Prusiner S, Stadtman ER (eds) The enzymes of glutamine metabolism. Academic Press, New York, pp 65–70
Magasanik B (1982) Genetic control of nitrogen assimilation in bacteria. Annu Rev Genet 16: 135–168
Matsuoka K, Kimura K (1986) Glutamate synthase from Bacillus subtilis PCI 219. J Biochem 99: 1087–1100
Meers JL, Tempest DW, Brown CM (1970) ‘Glutamine (amide): 2-oxoglutarate amino transferase oxido-reductase (NADP)’, an enzyme involved in the synthesis of glutamate by some bacteria. J Gen Microbiol 64: 187–194
Miller ES, Brenchley JE (1981) l-Methionine SR-sulfoximine-resistant glutamine synthetase from mutants of Salmonella typhimurium. J Biol Chem 256: 11307–11312
Miller RE, Stadtman ER (1972) Glutamate synthase from Escherichia coli. An iron sulfide flavoprotein. J Biol Chem 247: 7407–7419
Moat GA, Foster JW (1988) Microbial physiology, 2nd edn, John Wiley & Sons, New York Chichester Brisbane, pp 251–284
Phibbs JR, Bernlohr RW (1971) Purification, properties, and regulation of glutamic dehydrogenase of Bacillus licheniformis. J Bacteriol 106: 373–385
Schmidt CNG, Jervis L (1982) Partial purification and characterization of glutamate synthase from a thermophilic bacillus, J Gen Microbiol 128: 1713–1718
Schreier HJ, Bernlohr RW (1984) Purification and properties of glutamate synthase from Bacillus licheniformis. J Bacteriol 160: 591–599
Schreier HJ, Brown SW, Hirschi KD, Nomellini JF, Sonenshein AL (1989) Regulation of Bacillus subtilis glutamine synthetase gene expression by the product of the gln R gene. J Mol Biol 210: 51–63
Senior PJ (1975) Regulation of nitrogen metabolim in Escherichia coli and Klebsiella aerogenes: studies with the continuous-culture technique. J Bacteriol 123: 407–418
Sung H-C, Tachiki T, Kumagai H, Tochikura T (1984) Production and preparation of glutamate synthase from Brevibacterium flavum. J Ferment Technol 62: 371–376
Tachiki T, Wakisaka S, Kumagai H, Tochikura T (1981) Glutamine synthetase from Micrococcus glutamicus: effect of nitrogen sources in culture medium on enzyme formation and some properties of crystalline enzyme. Agric Biol Chem 45: 1487–1492
Tempest DW, Meers JL, Brown CM (1973) Glutamate synthetase (GOGAT); a key enzyme in the assimilation of ammonia by prokaryotic organisms. In: Prusiner S, Stadtman ER (eds) The enzymes of glutamine metabolism. Academic Press, New York, pp 167–182
Tochikura T, Sung H-C, Tachiki T, Kumagai H (1984) Occurrence of glutamate synthase in Brevibacterium flavum. Agric Biol Chem 48: 2149–2150
Tyler B (1978) Regulation of the assimilation of nitrogen compounds. In: Snell EE, Boyer PD, Meister A, Richardson CC (eds) Annual review of biochemistry, vol 47. Annual Review Inc., California, pp 1147–1162
Vandecasteele JP, Lemal J, Coudert M (1975) Pathways and regulation of glutamate synthesis in a Corynebacterium sp. over-producing glutamate. J Gen Microbiol 90: 178–180
Yamamoto I, Abe A, Saito H, Ishimoto M (1984) The pathway of ammonia assimilation in Bacteroides fragilis. J Gen Appl Microbiol 30: 499–508
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ertan, H. The effect of various culture conditions on the levels of ammonia assimilatory enzymes of Corynebacterium callunae . Arch. Microbiol. 158, 42–47 (1992). https://doi.org/10.1007/BF00249064
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00249064