Abstract
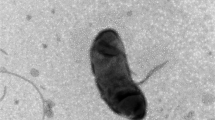
Gliding bacteria attached in masses to, and dominated the fermentation of, xylan powder in methanogenic and sulfidogenic enrichments from various freshwater sediments. Isolates of such bacteria were all gram-negative, slender rods (0.4×4-24 μm) that formed no endospores, microcysts or fruiting bodies. Representative strain XM3 was a mesophilic, aeroduric anaerobe that grew by fermentation of mono-, di-, and poly-saccharides (but not cellulose) in a mineral medium containing up to 3% NaCl. However, CO2/HCO sup-inf3 was required in media for consistent initiation of growth. Fermentation products included acetate, propionate, succinate, CO2, and H2. Xylan-grown cells had xylanase and various glycosidase activities that were mainly or almost entirely cell-associated, respectively. Strain XM3 was weakly catalase positive, but oxidase negative; it possessed sulphonolipids and carotenoid, but not flexirubin, pigments; and its total cellular fatty acids were dominated by C15:0 anteiso (75%), n (13%) and iso (2%) isomers. Strain XM3 had 45.5 mol% G+C in its DNA, and partial sequencing of its 16S rRNA placed XM3 within the Bacteroides-Flavobacterium phylogenetic group. Similar strains were isolated from marine sediments. Strain XM3 is herewith proposed as the type strain of the new species, Cytophaga xylanolytica. Results, which are discussed in terms of our current concept of the genus Cytophaga, suggest that the importance of C. xylanolytica in anaerobic biopolymer decomposition has not been fully appreciated.
Similar content being viewed by others
References
Biely P (1985) Microbial xylanolytic systems. Trends Biotechnol 3: 286–290
Breznak JA, Pankratz HS (1977) In situ morphology of the gut microbiota of wood-eating termites [Reticulitermes flavipes (Kollar) and Coptotermes formosanus Shiraki]. Appl Environ Microbiol 33. 406–426
Breznak JA, Switzer JM (1986) Acetate synthesis from H2 plus CO2 by termite gut microbes. Appl Environ Microbiol 52: 623–630
Breznak JA, Switzer JM, Seitz H-J (1988) Sporomusa termitida sp. nov., an H2/CO2-utilizing acetogen isolated from termites. Arch Microbiol 150: 282–288
Christensen P (1980) Description and taxonomic status of Cytophaga heparina (Pazya and Korn) comb. nov. (basionym: Flavobacterium heparinum Pazya and Korn 1956). Int J Syst Bacteriol 30: 473–475
Cord-Ruwisch R (1985) A quick method for the determination of dissolved and precipitated sulfides in cultures of sulfate-reducing bacteria. J Microbiol Methods 4: 33–36
Costilow R (1981) Biophysical factors in growth. In: Gerhardt P (ed) Manual of methods for general bacteriology, chapter 6. American Society for Microbiology, Washington, DC
Flossdort J (1983) A rapid method for the determination of the base composition of bacterial DNA. J Microbiol Methods 1: 305–311
Follett EAC, Webley DM (1965) An electron microscope study of the cell surface of Cytophaga johnsonii and some observations on related organisms. Antonie van Leeuwenhoek J Microbiol Serol 31: 361–382
Friedemann TE, Haugen GE (1943) Pyruvic acid. II. The determination of keto acids in blood and urine. J Biol Chem 147: 415–442
Godchaux WIII, Leadbetter ER (1983) Unusual sulfonolipids are characteristic of the Cytophaga-Flexibacter group. J Bacteriol 153: 1238–1246
Greve LC, Labavitch JM, Stack RA, Hungate RE (1984) Muralytic activities of Ruminococcus albus 8. Appl Environ Microbiol 47: 1141–1145
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW (eds) Methods in microbiology, vol. 5B. Academic Press, New York, pp 209–344
Hermann M, Noll KM, Wolfe RS (1986) Improved agar bottle plate for isolation of methanogens or other anaerobes in a defined gas atmosphere. Appl Environ Microbiol 51: 1124–1126
Lane DJ, Pace B, Olsen GJ, Stahl DA, Sogin ML, Pace NR (1985) Rapid determination of 16S ribosomal RNA sequences for phylogenetic analyses. Proc Natl Acad Sci USA 82: 6955–6959
Nicolaides N, Soukup VG, Ruth EC (1983) Mass spectrometric fragmentation patterns of the acetoxy and trimethylsilyl derivatives of all the positional isomers of the methyl hydroxypalmitates. Biomed Mass Spectrom 10: 441–449
Oyaizu H, Komagata K, Amemura A, Harada T (1982) A succinoglycan-decomposing bacterium, Cytophaga arvensicola sp. nov. J Gen Appl Microbiol 28: 369–388
Pfennig N, Wagener S (1986) An improved method of preparing wet mounts for photomicrographs of microorganisms. J Microbiol Methods 4: 303–306
Potrikus CJ, Breznak JA (1977) Nitrogen-fixing Enterobacter agglomerans isolated from guts of wood-eating termites. Appl Environ Microbiol 33: 392–399
Reichenbach H (1988) Gliding bacteria in biotechnology. In: Rehm H-J, Reed G (eds) Biotechnology, vol. 6B. Verlag Chemie, Weinheim pp 673–696
Reichenbach H (1989a) Order I. Cytophagales Leadbetter 1974, 99AL. In: Staley JT, Bryant MP, Pfennig N, Holt JG (eds) Bergey's manual of systematic bacteriology, vol. 3. Williams & Wilkins, Baltimore, pp 2011–2013
Reichenbach H (1989b) Family I. Cytophagaceae Stanier 1940, 630, AL emend. In: Staley JT, Bryant MP, Pfennig N, Holt JG (eds) Bergey's manual of systematic bacteriology, vol. 3. Williams & Wilkins, Baltimore. pp 2013–2015
Reichenbach H (1989c) Genus I. Cytophaga Winogradsky 1929, 577, AL, emend. In: Staley JT, Bryant MP, Pfennig N, Holt JG (eds) Bergey's manual of systematic bacteriology, vol. 3. Williams & Wilkins, Baltimore, pp 2015–2050
Reichenbach H, Kleinig H, Achenbach H (1974) The pigments of Flexibacter elegans: novel and chemosystematically useful compounds. Arch Microbiol 101: 131–144
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467
Schink B, Pfennig N (1982) Fermentation of trihydroxybenzenes by Pelobacter acidigallici gen. nov. sp. nov., a new strictly anaerobic, non-sporeforming bacterium. Arch Microbiol 133: 195–201
Skerman VBD, (1967) A guide to the identification of the genera of bacteria. Williams & Wilkins, Baltimore
Smibert RM, Krieg NR (1981) General characterization. In: Gerhardt P (ed) Manual of methods for general bacteriology, chapter 20. American Society for Microbiology, Washington, DC
Somogyi M (1952) Notes on sugar determination. J Biol Chem 195: 19–23
Umbreit WW, Burris RH, Stauffer JF (1957) Manometric techniques. Burgess, Minneapolis
Weisburg WG, Oyaizu Y, Oyaizu H, Woese CR (1985) Natural relationship between bacteroides and flavobacteria. J Bacteriol 164: 230–236
Widdel F, Kohring G-W, Mayer F (1983) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids. III. Charcterization of the filamentous gliding Desulfonema limicola gen. nov. sp. nov., and Desulfonema magnum sp. nov. Arch Microbiol 134: 286–294
Widdel F, Pfennig N (1984) Dissimilatory sulfate- or sulfur-reducing bacteria. In: Krieg NR, Holt JG (eds) Bergey's manual of systematic bacteriology, vol. 1. Williams & Wilkins, Baltimore, pp 663–679
Wilkie KCB (1983) Hemicellulose. Chemtech 13: 306–319
Woese CR (1987) Bacterial evolution. Microbiol Rev 51: 221–271
Zechmeister L (1962) Cis-trans isomeric carotenoids, vitamins A, and arylpolyenes. Springer, Wien
Author information
Authors and Affiliations
Additional information
Dedicated to Professor Norbert Pfennig, in whose laboratory this project was initiated and who recently retired after more than four decades of contributions to microbiology
Rights and permissions
About this article
Cite this article
Haack, S.K., Breznak, J.A. Cytophaga xylanolytica sp. nov., a xylan-degrading, anaerobic gliding bacterium. Arch. Microbiol. 159, 6–15 (1993). https://doi.org/10.1007/BF00244257
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00244257