Abstract
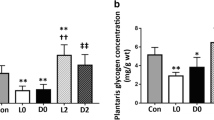
The aim of the present study was to examine the rate of glycogen mobilization during exercise and the rate of the postexercise glycogen replenishment in different muscle types [white (WG), and red (RG) gastrocnemius, soleus (S) and diaphragm (D)] in rats treated with triiodothyronine (T3, group T). Rats of the control group (C) were treated with saline. The animals were made to run on a treadmill set at 0° gradient and at a speed of 1200 m·h−1. The time taken to reach exhaustion in group C was 188 (SD 23) min, whereas in group T, it was only 63 (SD 12) min. The content of glycogen in all muscles of the rats from group T at rest and during exercise was significantly lower than in group C at each corresponding time. At exhaustion, the glycogen content was in WG(C) 34.79 (SD 4.65), (T) 20.10 (SD 4.10); in RG(C) 22.82 (SD 4.66), (T) 16.50 (SD 2.00); in S(C) 14.85 (SD 2.48), (T) 11.90 (SD 2.93); in D(C) 18.18 (SD 3.49), (T) 7.54 (SD 3.36) (μmol of glucosyl units·g−1). The amount of glycogen mobilized during exhausting exercise in RG, S and D was similar in both groups whereas in WG it was much higher in rats of group T than in group C. The concentration of glycogen returned to pre-exercise values in each muscle 3 h after exercise. The net amount of glycogen resynthetized during 3 h of recovery depended on the muscle type. It was in WG(C) 3.30, (T) 18.03; in RG(C) 21.34, (T) 25.88, in S(C) 34.00, (T) 17.68, and in D(C) 17.25, (T) 12.22 μmol of glucosyl units·g−1 (each number represents the difference between the means). It concluded that treatment with T3 markedly affects this exercise-induced metabolism of glycogen in each muscle type.
From our study it is suggested that low muscle glycogen content may contribute to a reduction in exercise performance in hyperthyroidism.
Similar content being viewed by others
References
Ariano MA, Armstrong RB, Edgerton VR (1973) Hindlimb muscle fiber population of five mammals. J Histochem Cytochem 21:51–55
Armstrong RG, Saubert WC, Sembrowich WL, Shepherd RE, Gollnick PD (1974) Glycogen depletion in rat skeletal muscle fibers at different intensities and durations of exercise. Pflügers Arch 352:243–256
Baldwin KM, Reitman JS, Terjung RL, Winder WW, Holloszy JO (1973) Substrate depletion in different types of muscle and in liver during prolonged running. Am J Physiol 225:1045–1050
Battarbee HD (1974) The effects of thyroid state on rat liver glucose-6-phosphatase activity and glycogen content. Proc Soc Exp Biol Med 147:337–343
Brzezińska Z, Kaciuba-Uściłko H (1979) Low muscle and liver glycogen contents in dogs treated with thyroid hormones. Horm Metab Res 11:675–678
Carroll NV, Longley RW, Roe JH (1956) The determination of glycogen in liver and muscle by use of anthrone reagent. J Biol Chem 220:583–593
Coulee RK, Hickson RC, Winder WW, Hagberg JM, Holloszy JO (1978) Regulation of glycogen resynthesis in muscles of rats following exercise. Am J Physiol 235:R145-R150
Costill DL, Coyle E, Daisky G, Evans W, Fink W, Hoopes D (1977) Effects of elevated plasma FFA and insulin on muscle glycogen usage during exercise. J Appl Physiol 43:695–699
Dimitriadis GD, Leighton B, Vlachonikolis IG, Party-Billings M, Challiss RAJ, West D, Newsholme E (1988) Effects of hyperthyroidism on the sensitivity of glycolysis and glycogen synthesis to insulin in the soleus muscle of the rat. Biochem J 253:87–92
Dubaniewicz A, Kaciuba-Uściłko H, Nazar K, Budohoski L (1989) Sensitivity of the soleus muscle to insulin in resting and exercing rats with experimental hypo- and hyper-thyroidism. Biochem J 263:243–247
Duncombe WG (1964) The colorimetric micro-determination of nonesterified fatty acids in plasma. Clin Chim Acta 9:122–125
Gauthier GF, Padykula HA (1966) Cytological studies of fiber type in skeletal muscle. A comparative study of the mammalian diaphragm. J Cell Biol 28:333–354
Gollnick PD, Piehl K, Saltin B (1974) Selective glycogen depletion pattern in human muscle fibers after exercise of varying intensity and at varying pedalling rats. J Physiol 241:45–57
Górski J (1978) Exercise-induced changes of reactivity of different types of muscle on glycogenolytic effect of adrenaline. Pflügers Arch 373:1–7
Górski J, Krawczuk I, Górska M, Rutkiewicz J (1991) Inhibition of glycogenesis in rat muscles partially depleted of glycogen. Am J Physiol 261:C305-C309
Hickson RC, Rennie MJ, Coulee RK, Winder WW, Holloszy JO (1977) Effect of increased plasma fatty acids on glycogen utilization and endurance. J Appl Physiol 43:829–833
Krause EG, Wollenberger A (1968) Influence of thyroid state on enzymes of glycogen metabolism in rat skeletal muscle. Acta Biol Med Ger 21:615–624
Kruk B, Brzezińska Z, Kaciuba-Uściłko H, Nazar K (1988) Thyroid hormones and muscle metabolism in dogs. Horm Metab Res 20:620–623
Kubista V, Kubistova J, Pette D (1971) Thyroid hormone induced changes in the enzyme activity pattern of energy supplying metabolism of fast (white), slow (red) and heart muscle of the rat. Eur J Biochem 18:553–560
Leighton B, Dimitriadis GD, Parry-Billings M, Bond J, Kenys P, Newsholme EA (1990) Thyroid hormone analogue SKF L-94901: effects on amino acid and carbohydrate metabolism in rat skeletal muscle in vitro. Biochem Pharmacol 40:1161–1164
Pawłowski Z (1969) An introduction to mathematical statistics. PZWL, Warsaw.
Rennie MJ, Winder WW, Holloszy JO (1976) A sparing effect of increased plasma fatty acids on muscle and liver glycogen content in the exercising rat. Biochem J 156:647–655
Saltin B, Gollnick PD (1988) Fuel for muscular exercise: role of carbohydrate In: Horton ES, Terjung RL (eds) Exercise, nutrition and energy metabolism. Macmillan, New York, pp 44–71
Sandler MP, Robinson RP, Rabin D, Lacy WW, Abumrad NN (1983) The effect of thyroid hormones on gluconeogenesis and forearm metabolism in man. J Clin Endocrinol Metab 56:479–485
Shoji S (1991) Effect of thyroxine on basal and insulin stimulated glucose uptake by fast and slow skeletal muscles of rats. Comp Biochem Physiol 100A:343–345
Sullivan TE, Armstrong RB (1978) Rat locomotory muscle fiber activity during trotting and galloping. J Appl Physiol Respir Environ Exerc Physiol 44:358–363
Terjung RL, Baldwin KM, Winder WW, Holloszy JO (1974) Glycogen repletion in different types of muscle and in liver after exhausting exercise. Am J Physiol 226:1387–1391
Wiles CM, Young A, Jones DA, Edwards RHT (1979) Muscle relaxation rate, fibre-type composition and energy turnover in hyper-and hypo-thyroid patients. Clin Sci 57:375–384
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kudelska, G., Górski, J., Swiątecka, J. et al. Effect of exercise on glycogen metabolism in muscles of triiodothyronine-treated rats. Eur J Appl Physiol 72, 496–501 (1996). https://doi.org/10.1007/BF00242281
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00242281