Abstract
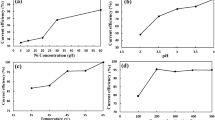
The effect of chromate ion and its interaction with antimony and glue on zinc electrowinning from acid sulphate electrolyte were studied. During electrolysis at 430 A m−2 and 45°C, the chromate ion was found to polarize the cathodic reaction resulting in a refined zinc deposit having a morphology featuring a vertical orientation of the zinc platelets. Although chromium did not codeposit, the current efficiency for zinc deposition was dramatically decreased in the presence of > 1000 mg dm−3. This effect became more pronounced when the electrolyte also contained additives such as antimony and glue.
Similar content being viewed by others
References
R. C. Kerby and C. J. Krauss, ‘Lead-Zinc-Tin `80’ (edited by J. M. Cigan, T. S. Mackey and T. J. O'Keefe), TMS-AIME, New York (1979) p. 187.
M. K. Gupta, B. L. Gupta and R. Raghavan, Hydrometall. 22 (1989) 379.
I. Nirdosh, R. K. Kalia and S. V. Muthuswami, ibid. 20 (1988) 203.
J. F. Collins and A. M. Wolsky, A ‘White Paper’ prepared for the Waste Products Utilization Office of Industrial Programs US Department of Energy (1987).
D. J. Mackinnon, J. M. Brannen and P. L. Fenn, J. Appl. Electrochem. 17 (1987) 1129.
I. W. Wark, ibid. 9 (1979) 721.
D. R. Fosnacht and T. J. O'Keefe, ibid. 10 (1980) 495.
J.-C. Lin, Proceedings of ROC and ROK Bilateral Workshop on Mineral Beneficiation, Hydrometallurgy and Environmental Controls, Mining Research and Service Organization, Taiwan, ROC (1980) pp. 217–230.
S.-L. Tsai and J.-C. Lin, Bull. CIMME: Mining & Metallurgy 34(4) (1990) 116.
A. C. Scote, R. M. Pitblado, G. W. Barton and A. R. Ault, J. Appl. Electrochem. 18 (1988) 120.
B. A. Lamping and T. J. O'Keefe, Met. Trans., 7B (1976) 551.
R. C. Kerby, H. E. Jackson, T. J. O'Keefe, and Yar-Ming Wang, Met. Trans. B, 8B (1977) 661.
D. J. Mackinnon, R. M. Morrison, J. E. Mouland and P. E. Warren, J. Appl. Electrochem. 20 (1990) 728.
I. Epelboin, M. Ksouri and R. Wiart, J. Electrochem. Soc. 122 (1975) 1206.
M. Pourbaix, ‘Atlas of Electrochemical Equilibria’, Pergamon Press, New York (1966) p. 261.
B. N. Popov, R. E. White, D. Slavkov and Z. Koneska, J. Electrochem. Soc. 139 (1992) 91.
G. Isserlis, in ‘Industrial Electrochemical Processes’ (edited by A. T. Kuhn) Elsevier, New York (1971) p. 355.
A. Radwan, A. El-Kiar, H. A. Farag, and G. H. Sedahmed, J. Appl. Electrochem. 22 (1992) 1161.
J. P. Hoare, A. H. Holden and M. A. LaBoda, Plat. Surf. Finish 67(3) (1980) 42.
J. P. Hoare, J. Electrochem. Soc. 130 (1983) 1475.
R. R. Lloyed, W. T. Rawles and R. G. Feeney, Trans. Electrochem. Soc. 89 (1946) 443.
J. S. Lee and T. Sekine, Denki Kagaku 44 (1976) 176.
H. H. Fukubayashi, T. J. O'Keefe and W. C. Clinton, US Bureau of Mines, Report of Investigations (1974) No. 7966.
F. A. Cotton and G. Wilkinson, ‘Advanced Inorganic Chemistry’, 4th edn., John Wiley & Sons, New York (1980) p. 471.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lin, JC., Tsai, SL. Effect of chromate ion on zinc electrowinning from acid sulphate electrolyte. J Appl Electrochem 24, 1044–1051 (1994). https://doi.org/10.1007/BF00241197
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00241197