Abstract
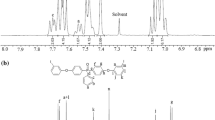
Anodic oxidation of p-methylanisole (1a) in a solution of aqueous 0.05 m H2SO4/acetone cosolvent (1 : 1, v/v) on platinum gives 5,4′-dimethyl-2-methoxydiphenylether (3), a compound hitherto unexplored, in 50% yield at a temperature below 15°C. Polymer products are formed in significant amounts (up to 60%) besides the diphenylether derivative (3) when the electrolytic oxidation proceeds in a solution containing AcOH as cosolvent at room temperature. The reaction mechanism includes both side chain anodic oxidation and ring 6e oxidation leading to a reactive intermediate, probably 3-methyl-5-methoxy-1,2-benzoquinone (5), whose polymeric products were also isolated.
Similar content being viewed by others
References
F. Fichter and H. Ris, Helv. Chim. Acta 7 (1924) 803.
D. Degner and H. Hannebaum, BASF Pat., C.A. 93: P 566 963 (l980).
I. Nishiguchi and T. Hirashima, J. Org. Chem. 50 (1985) 539.
S. Torii, H. Tanaka, T. Ikoguchi and S. Nakane, ibid. 47 (1982) 1647.
B. Wermeckes and F. Beck, Chem. Ber. 118 (1985) 3771.
J. N. Ashley, J. Chem. Soc. (1937) 1471.
C. L. Chen, W. J. Connors and W. M. Shinker, J. Org. Chem. 34 (1969) 2966.
V. D. Parker, A. Nilsson and A. Ronlan, J. Chem. Soc. (Perkin I) (1973) 23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hlavatý, J. 5,4′-dimethyl-2-methoxydiphenylether: a new product from the anodic oxidation of 4-methylanisole. J Appl Electrochem 24, 989–992 (1994). https://doi.org/10.1007/BF00241189
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00241189