Summary
Purkinje cell (PC), activity in the flocculus of trained monkeys was recorded during: 1) Vestibular stimulation in darkness. 2) Suppression of the vestibulo-ocular reflex (VOR-supp) by fixation of a small light spot stationary with respect to the monkey. 3) Visual-vestibular conflict (i.e. the visual surround moves together with the monkey during vestibular stimulation), which leads to attenuation or suppression of vestibular nystagmus. 4) Smooth pursuit eye movements. 5) Optokinetic nystagmus (OKN). 6) Suppression of nystagmus during optokinetic stimulation (OKN-supp) by fixation of a small light spot; whereby stimulus velocity corresponds then to image slip velocity.
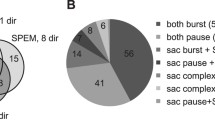
Results were obtained from PCs, which were activated with VOR-supp during rotation to the ipsilateral side. The same PCs were also modulated during smooth pursuit and visual-vestibular conflict. No tonic modulation during constant velocity OKN occurred with slow-phase nystagmus velocities below 40–60 deg/s. Tonic responses were only seen at higher nystagmus velocities. Transient activity changes appeared at the beginning and end of optokinetic stimulation. PCs were not modulated by image slip velocity during OKN-supp.
The results show that in primates the same population of floccular PCs is involved in different mechanisms of visual-vestibular interaction and that smooth pursuit and certain components of OKN slow-phase velocity share the same neural pathway. It is argued that the activity of these neurons can neither be related strictly to gaze, eye or image slip velocity; instead, their activity pattern can be best interpreted by assuming a modulation, which is complementary to that of central vestibular neurons of the vestibular nuclei, in the control of slow eye movements.
Similar content being viewed by others
References
Bell CC, Grimm RJ (1969) Discharge properties of Purkinje cells recorded on single and double microelectrodes. J Neurophysiol 32: 1044–1055
Buettner UW, Büttner U (1979) Vestibular nuclei activity in the alert monkey during suppression of vestibular and optokinetic nystagmus. Exp Brain Res 37: 581–593
Buettner UW, Büttner U, Henn V (1978) Transfer characteristics of neurons in the vestibular nuclei of the alert monkey. J Neurophysiol 41: 1614–1628
Büttner U, Boyle R, Schreiter U (1983a) Vestibular nuclei activity in the alert monkey during constant velocity and sinusoidal optokinetic stimulation. Soc Neurosci Abstr 9: 315
Büttner U, Meienberg O, Schimmelpfennig B (1983b) The effect of central retinal lesions on optokinetic nystagmus in the monkey. Exp Brain Res 52: 248–256
Büttner U, Waespe W, Henn V (1981) The role of the cerebellum and the vestibular system in the generation of slow conjugate eye movements. In: Huber A, Klein D (eds) Neurogenetics and neuro-ophthalmology. Elsevier/North-Holland Biomedical Press, Amsterdam, pp 89–102
Cohen B, Matsuo V, Raphan T (1977) Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic afternystagmus. J Physiol (Lond) 270: 321–344
Cohen B, Suzuki J, Raphan T, Matsuo V, de Jong V (1982) Selective labyrinthine lesions and nystagmus induced by rotation about off-vertical axis. In: Lennerstrand G, Keller E, Zee DS (eds) Functional basis of ocular motility disorders. Pergamon Press, Oxford, pp 337–346
Dichgans J, v. Reutern GM, Römmelt U (1978) Impaired suppression of vestibular nystagmus by fixation in cerebellar and non-cerebellar patients. Arch Psychiat Nervenkr 226: 183–199
Haines DE (1977) Cerebellar corticonuclear and corticovestibular fibers of the flocculonodular lobe in a prosimian primate (Galago senegalensis). J Comp Neurol 174: 607–630
Keller EL, Daniels PD (1975) Oculomotor related interaction of vestibular nucleus cells in alert monkey. Exp Neurol 46: 187–198
Lisberger SG, Fuchs AF (1978) Role of primate flocculus during rapid behavioral modification of vestibuloocular reflex. I. Purkinje cell activity during visually guided horizontal smooth-pursuit eye movements and passive head rotation. J Neurophysiol 41: 733–763
Lisberger SG, Miles FA, Optican LM, Eighmey BB (1981) Optokinetic response in monkey: Underlying mechanisms and their sensitivity to long-term adaptive changes in vestibuloocular reflex. J Neurophysiol 45: 869–890
Miles FA, Fuller JH, Braitman DG, Dow BM (1980) Long-term adaptive changes in primate vestibuloocular reflex. III. Electrophysiological observations in flocculus of normal monkeys. J Neurophysiol 43: 1437–1476
Raphan T, Cohen B (1981) The role of integration in oculomotor control. In: Zuber B (ed) Models of oculomotor behavior and control. CRC Press Inc., West Palm Beach, Fla, pp 91–109
Robinson DA (1981) Control of eye movements. In: Brooks VB (ed) Handbook of physiology, section I: The nervous system, vol II, part 2. American Physiological Society, Bethesda, Md, pp 1275–1320
Takemori S, Cohen B (1974) Loss of visual suppression of vestibular nystagmus after flocculus lesions. Brain Res 72: 213–224
Waespe W, Büttner U, Henn V (1981) Visual-vestibular interaction in the flocculus of the alert monkey. I. Input activity. Exp Brain Res 43: 337–348
Waespe W, Cohen B, Raphan T (1983) Role of the flocculus in optokinetic nystagmus and visual-vestibular interactions: Effects of flocculectomy. Exp Brain Res 50: 9–33
Waespe W, Henn V (1977a) Neuronal activity in the vestibular nuclei of the alert monkey during vestibular and optokinetic stimulation. Exp Brain Res 27: 523–538
Waespe W, Henn V (1977b) Vestibular nuclei activity during optokinetic afternystagmus (OKAN) in the alert monkey. Exp Brain Res 30: 323–330
Waespe W, Henn V (1978) Conflicting visual-vestibular stimuli and vestibular nucleus activity in alert monkeys. Exp Brain Res 33: 203–211
Waespe W, Henn V (1979) The velocity response of vestibular nucleus neurons during vestibular, visual and combined angular acceleration. Exp Brain Res 37: 337–347
Waespe W, Henn V (1981) Visual-vestibular interaction in the flocculus of the alert monkey. II. Purkinje cell activity. Exp Brain Res 43: 349–360
Waespe W, Henn V (1984) Cooperative functions of central vestibular neurons and floccular P-cells in the control of nystagmus slow phase velocity: Single cell recordings and lesion studies in the monkey. Rev Oculomotor Res 1 (in press)
Waespe W, Rudinger D, Wolfensberger M, Henn V Floccular Purkinje cell activity after bilateral labyrinthectomy during optokinetic nystagmus (OKN) and smooth pursuit eye movements in primates (in preparation)
Wurtz RH (1969) Visual receptive fields of striate cortex neurons in awake monkeys. J Neurophysiol 32: 727–742
Zee DS, Yamazaki A, Butler PH, Gücer G (1981) Effects of ablation of flocculus and paraflocculus on eye movements in primates. J Neurophysiol 46: 878–899
Author information
Authors and Affiliations
Additional information
Supported by Swiss National Foundation for Scientific Research 3.343-2.78, and Deutsche Forschungsgemeinschaft, SFB 200, A2
Rights and permissions
About this article
Cite this article
Büttner, U., Waespe, W. Purkinje cell activity in the primate flocculus during optokinetic stimulation, smooth pursuit eye movements and VOR-suppression. Exp Brain Res 55, 97–104 (1984). https://doi.org/10.1007/BF00240502
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00240502