Summary
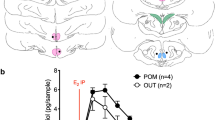
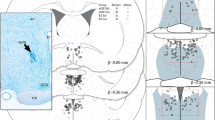
The purpose of this study was to determine the specificity of the response of medial preoptic-septal neurons (mPOA-S) to microelectrophoresed 17β-estradiol hemisuccinate (17βE2S). In vitro studies were conducted initially to determine the release of the labeled 17βE2S from multibarrel glass micropipettes. Subsequently, an isomer of 17βE2S, 17α-estradiol hemisuccinate (17αE2S), was synthesized and purified. Thirty-six mPOA-S neurons from normal cycling female rats were tested with both 17βE2S and 17αE2S. Twelve of these units responded with inhibition to 17βE2S, while none responded to 17αE2S. Furthermore, fifty extrahypothalamic (cortical, hippocampal, thalamic) neurons were tested with 17βE2S. The majority (N = 45) showed no response, three showed excitation and two inhibition to the microelectrophoresed steroid ester. These findings suggest that a specific receptor mechanism is responsible for the changes in mPOA-S unit activity, and that these effects may be important in the regulation of reproductive events.
Similar content being viewed by others
References
Abraham, G.E., Grover, P.K.: Covalent linkage of steroid hormones to protein carriers for use in radioimmunoassay. In: Principles of Competitive Protein-Binding Assays, Chapter VI-B (eds. W.O. Odell and W.H. Daughaday), pp. 140–157. Philadelphia: Lippincott Publishing 1971
Anderson, C.H., Greenwald, G.S.: Autoradiographic analysis of estradiol uptake in the brain and pituitary of the female rat. Endocrinology 85, 1160–1165 (1969)
DeGroot, J.: The rat hypothalamus in stereotaxic coordinates. J. comp. Neurol. 113, 389–400 (1959)
Eisenfeld, A.J., Axelrod, J.: Selectivity of estrogen distribution in tissues. J. Pharmacol. exp. Ther. 150, 469–475 (1965)
Hoffer, B.J., Neff, N.H., Siggins, G.R.: Microelectrophoretic release of norepinephrine from micropipettes. Neuropharmacology 10, 175–180 (1971)
Kato, J.: In vitro uptake of tritiated oestradiol by the rat anterior hypothalamus during the oestrous cycle. Acta endocr. (Kbh.) 63, 577–584 (1970a)
Kato, J.: In vitro uptake of tritiated oestradiol by the anterior hypothalamus and hypophysis of the rat. Acta endocr. (Kbh.) 64, 687–704 (1970b)
Kato, J.: Localization of oestradiol receptors in the rat hypothalamus. Acta endocr. (Kbh.) 72, 663–670 (1973)
Kato, J., Villee, C.A.: Preferential uptake of oestradiol by the anterior hypothalamus of the rat. Endocrinology 80, 567–575 (1967a)
Kato, J., Villee, C.A.: Factors affecting uptake of estradiol-6,7-3H by the hypophysis and hypothalamus. Endocrinology 80, 1113–1138 (1967b)
Kelly, M.J., Dudley, C.A., Moss, R. L.: Identification of estrogen-sensitive neurons in the preoptic-septal area of the normal cyclic female rat. Soc. Neurosci. 709A (1975)
Kelly, M.J., Moss, R.L.: Quantitative evaluation and determination of the biological potency of iontophoretically applied luteinizing hormone-releasing hormone (LRH). Neuropharmacology 15, 325–328 (1976)
Kelly, M.J., Moss, R.L., Dudley, C.A.: Differential sensitivity of preoptic-septal neurons to microelectrophoresed estrogen during the estrous cycle. Brain Res. 114, 152–157 (1976)
Kelly, M.J., Moss, R.L., Dudley, C.A.: The effects of microelectrophoretically applied estrogen, cortisol and acetylcholine on medial preoptic-septal unit activity throughout the estrous cycle of the female rat. Exp. Brain Res. 30, 53–64 (1977)
Krnjević, K.: Glutamate and α-Aminobutyric acid in brain. Nature (Lond.) 228, 119–124 (1970)
Livingston, J.N., Lockwood, D.H.: Effect of glucocorticoids on the glucose transport system of isolated fat cells. J. biol. Chem. 250, 8353–8360 (1975)
Mandelbrod, J., Feldman, S., Weiman, R.: Inhibition of firing is the primary effect of microelectrophoresis of cortisol to units in the rat tuberal hypothalamus. Brain Res. 80, 303–315 (1974)
McCann, S.M.: Regulation of secretion of follicle-stimulating hormone and luteinizing hormone. In: Handbook of Physiology, Section 7: Endocrinology, Vol. IV. The Pituitary Gland, Part 2 (eds. R.O. Greep and E.B. Astwood), pp. 489–518. Washington, D.C.: American Physiological Society 1974
McEwen, B., Pfaff, D.W.: Factors influencing sex hormone uptake by rat brain regions. I. Effects of neonatal treatment, hypophysectomy and competing steroids on estradiol uptake. Brain Res. 21, 1–16 (1970)
Pfaff, D.W.: Autoradiographic localization of radioactivity in rat brain after injection of tritiated sex hormones. Science 161, 1355–1356 (1968)
Pfaff, D.W., Keiner, M.: Atlas of estradiol-concentrating cells in the central nervous system of the female rat. J. comp. Neurol. 151, 121–158 (1973)
Siegel, S.: Nonparametric Statistics for the Behavioral Sciences, pp. 166–172. New York: McGraw-Hill 1956
Stumpf, W.E.: Estradiol-concentrating neurons: topography in the hypothalamus by dry-mount autoradiography. Science 162, 1001–1003 (1968)
Wilks, S.S.: Mathematical Statistics, Chapter 13.4, pp. 408–411. New York: J. Wiley 1962
Yagi, K.: Effects of estrogen on the unit activity of the rat hypothalamus. J. Physiol. Soc. Japan. 32, 692–693 (1970)
Yagi, K.: Changes in firing rates of single preoptic and hypothalamic units following an intravenous administration of estrogen in the castrated female rat. Brain Res. 53, 343–352 (1973)
Yagi, K., Sawaki, Y.: Feedback of estrogen in the hypothalamic control of gonadotropin secretion. In: Neuroendocrine Control (eds. K. Yagi and S. Yoshida), pp. 297–325. Japan: University of Tokyo Press 1973
Zigmond, R.E., McEwen, B.S.: Selective retention of oestradiol by cell nuclei in specific brain regions of the ovariectomized rat. J. Neurochem. 17, 889–899 (1970)
Author information
Authors and Affiliations
Additional information
Supported by NIH Grant NS10434-END, awarded to R.L. Moss
Presently an NIH Postdoctoral Fellow at Max-Planck Institute for Biophysical Chemistry, Göttingen, West Germany
Recipient of an USPHS Career Development Award No. HD00146
Rights and permissions
About this article
Cite this article
Kelly, M.J., Moss, R.L., Dudley, C.A. et al. The specificity of the response of preoptic-septal area neurons to estrogen: 17α-estradiol versus 17β-estradiol and the response of extrahypothalamic neurons. Exp Brain Res 30, 43–52 (1977). https://doi.org/10.1007/BF00237857
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00237857