Summary
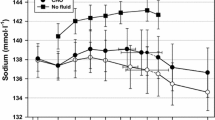
The effects of carbohydrate loading on relative stress responses of eight male subjects performing intermittent leg exercise at 80% maximum oxygen consumption during headout immersion in 25°C water were tested. Carbohydrate loading increased the number of work cycles completed, with less physical stress compared with that completed following the control diet period. Pre-exercise serum cortisol values were similar on both diets prior to exercise but following exercise control values were greater (1152, 94 vs 858, 77 nmol l−1; mean, SEM). Chromium losses, which have been shown to correlate with stress, were lower during the carbohydrate loading period, 8.6, 1.3 vs 12.4, 2.0 ng h−1, and were correlated with post-exercise serum cortisol. Urinary zinc losses were also lower during carbohydrate loading, while urinary losses of potassium, magnesium and calcium remained constant. Insulin values decreased similarly following exercise in both groups and were not altered by carbohydrate loading. These data demonstrate that carbohydrate loading increases immersion exercise output with less stress as determined by serum cortisol and urinary chromium losses.
Similar content being viewed by others
References
Anderson RA (1987) Chromium. In: Mertz W (ed) Trace elements in human and animal nutrition. Academic Press, Orlando, Fla., pp 225–244
Anderson RA, Guttman HN (1988) Trace minerals and exercise. In: Terjung R, Horton ES (eds) Exercise, nutrition and energy metabolism. Macmillan, New York, pp 180–195
Anderson RA, Kozlovsky AS (1985) Chromium intake, absorption and excretion of subjects consuming self-selected diets. Am J Clin Nutr 41:1177–1183
Anderson RA, Polansky MM, Bryden NA, Roginsky EE, Patterson KY, Reamer DC (1982) Effect of exercise (running) on serum glucose, insulin, glucagon and chromium excretion. Diabetes 31:212–216
Anderson RA, Polansky MM, Bryden NA, Patterson KY, Veillon C, Glinsmann WH (1983) Effects of chromium supplementation on urinary Cr excretion of human subjects and correlation of Cr excretion with selected clinical parameters. J Nutr 113:276–281
Anderson RA, Polansky MM, Bryden NA (1984) Strenuous running: acute effects on chromium, copper, zinc and selected clinical variables in urine and serum of male runners. Biol Trace Elements Res 6:327–336
Anderson RA, Polansky MM, Bryden NA, Bhathena SJ, Canary J (1987) Effects of supplemental chromium on patients with symptoms of reactive hypoglycemia. Metabolism 36:351–355
Anderson RA, Bryden NA, Polansky MM, Deuster PA (1988a) Exercise effects on chromium excretion of trained and untrained men consuming a constant diet. J Appl Physiol 64:249–252
Anderson RA, Borel JS, Polansky MM, Bryden NA, Majerus TC, Moser PB (1988b) Chromium intake and excretion of patients receiving total parenteral nutrition: effects of supplemental chromium. J Trace Elements Exp Med 1:9–18
Anderson RA, Bryden NA, Polansky MM, Reiser S (1990) Urinary chromium excretion and insulinogenic properties of carbohydrates. Am J Clin Nutr 51:864–868
Bergstrom J, Hultman E (1967) A study of the glycogen metabolism during exercise in man. Scand J Clin Lab Invest 19:218–228
Bergstrom J, Hermansen L, Hultman E (1967) Diet, muscle glycogen and physical performance. Acta Physiol Scand 71:140–150
Borel JS, Majerus TC, Polansky MM, Moser PB, Anderson RA (1984) Chromium intake and urinary chromium excretion of trauma patients. Biol Trace Elements Res 6:317–326
Bray JT, Van Rij AM, Hall MT, Pories WJ (1979) Analytical and sampling factors affecting urinary zinc analyses. In: Hemphill DD (ed) Trace substances in environmental health, vol XIII. Columbia, Mo., pp 163–171
Christensen EH, Hansen O (1939) Respiratorischer Quotient und O2-Aufnahme. Scand Arch Physiol 81:180–189
Consolazio CF, Nelson RA, Matoush LR, Hughes RC, Urone P (1964) The trace element mineral losses in sweat. (Report no. 284) US Army Medical Research and Nutrition Laboratory, Denver, Colo.
Costill DL, Sherman WM, Fink WJ, Maresh C, Witten M, Miller JM (1981) The role of dietary carbohydrates in muscle glycogen resynthesis after strenuous running. Am J Clin Nutr 34:1831–1836
Davies CT, Few JD (1973) Effects of exercise on adrenocortical function. J Appl Physiol 35:887–891
Few JD (1974) Effect of exercise on the secretion and metabolism of cortisol in man. J Endocrinol 62:341–353
Fields M, Ferretti RJ, Smith JC, Reiser S (1984) The interaction of type of dietary carbohydrates with copper deficiency. Am J Clin Nutr 39:289–294
Forgac MT (1979) Carbohydrate loading — a review. J Am Diet Assoc 75:42–45
Greenleaf JE, Shvartz E, Kravik S, Keil LC (1980) Fluid shifts and endocrine responses during chair rest and water immersion in man. J Appl Physiol 60:176–183
Hultman E (1967) Studies on muscle metabolism of glycogen and active phosphate in man with special reference to exercise and diet. Scand J Clin Lab Invest 19:94
Karlsson J, Saltin B (1981) Diet, muscle glycogen and endurance performance. J Appl Physiol 31:203–226
Kozlovsky AS, Moser PB, Reiser S, Anderson RA (1986) Effects of diets high in simple sugars on urinary chromium losses. Metabolism 35:515–518
Kuoppasalmi K, Naveri H, Harkonen M, Adlercreutz H (1980) Plasma cortisol, androstinedione, testosterone and lutenizing hormone in running exercise of different intensities. Scand J Clin Lab Invest 40:403–409
Miller-Ihli NJ, Wolf WR (1986) Characterization of a diet reference material for 17 elements. Anal Chem 58:3225–3230
Pekarek RS, Hauer EC, Rayfield EJ, Wannemacher RW, Beisel WR (1975) Relationship between serum chromium concentrations and glucose utilization in normal and infected subjects. Diabetes 24:350–353
Sherman WM, Costill DL, Fink WJ, Miller JM (1981) Effect of exercise-diet manipulation on muscle glycogen and its subsequent utilization during performance. Int J Sports Med 2:114–118
Thorp JW, Mittleman KD, Haberman KJ, House JF, Doubt TJ (1990) Work enhancement and thermal changes during intermittent work in cool water after carbohydrate loading. Naval Medical Research Institute, Washington, DC, pp 80–114
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Anderson, R.A., Bryden, N.A., Polansky, M.M. et al. Effects of carbohydrate loading and underwater exercise on circulating cortisol, insulin and urinary losses of chromium and zinc. Eur J Appl Physiol 63, 146–150 (1991). https://doi.org/10.1007/BF00235185
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00235185